��Ŀ����
����Ŀ����ͭ��(��Ҫ�ɷ�ΪCuFeS2)������ͭ�����������ԭ�ϡ��ش���������:
��1����̬Cuԭ�ӵļ۵����Ų�ʽΪ________
��2����ԭ�ӽṹ�Ƕȷ���,��һ������I1(Fe)��I1(Cu)�Ĺ�ϵ��:I1(Fe)____I1(Cu)(�>��<"��=��)
��3��Ѫ����������(C4H5N)����Ҫ�����Ѫ����(��Fe2+)����������ȱ����ƶѪ�����Ժ�Ѫ���صĽṹ����ͼ:

����֪�����еĸ���ԭ�Ӿ���ͬһƽ���ڣ�������������Nԭ�ӵ��ӻ�����Ϊ_______
��1mol���������������ĦҼ�����Ϊ____���������еĴ�м�����![]() ��ʾ������m���������γɴ�м���ԭ����,n���������γɴ�м��ĵ�����,�����Ի��еĴ�м�Ӧ��ʾΪ_____��
��ʾ������m���������γɴ�м���ԭ����,n���������γɴ�м��ĵ�����,�����Ի��еĴ�м�Ӧ��ʾΪ_____��
��C��N��O����Ԫ�صļ��⻯���У��е��ɵ͵��ߵ�˳��Ϊ________(�ѧʽ)��
��ѪҺ�е�O2����Ѫ�������������γɵ�Ѫ�쵰�������͵�,��Ѫ�쵰���е�Fe2+��O2��ͨ��_____�����ϡ�
��4����ͭ��ұ��ͭʱ������SO2�ɾ���SO2![]() SO3
SO3![]() H2SO4;���γ����ꡣSO2�Ŀռ乹 ��Ϊ________��H2SO4������ǿ��H2SO3��ԭ����____________
H2SO4;���γ����ꡣSO2�Ŀռ乹 ��Ϊ________��H2SO4������ǿ��H2SO3��ԭ����____________
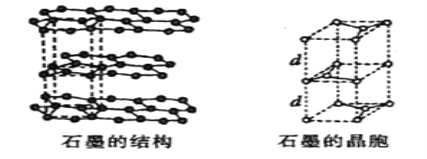
��5����ʯī���缫������ͭ����Ƶ�����ͭ��Һ�͵�����ʯī�ľ���ṹ����ͼ��ʾ,���߹��ճ������侧������ʯī�����к�̼ԭ����Ϊ____������֪ʯī���ܶ�Ϊ��g/cm3,C-C���ļ���Ϊrcm,�谢���ӵ�������ֵΪNA,��ʯī����IJ���d=______cm��
���𰸡� 3d104s1 �� sp2 10NA ![]() CH4��NH3��H2O ��λ�� V�� SO2(OH)2(��H2SO4)��S�Ļ��ϼ�Ϊ+6��S��������ǿ��SO(OH)2����H2SO3���е�S��ʹ�ǻ���O��H��Ĺ��õ��ӶԸ���ƫ��Oԭ�ӣ��ǻ����������H+��������H2SO4ǿ��H2SO3 4
CH4��NH3��H2O ��λ�� V�� SO2(OH)2(��H2SO4)��S�Ļ��ϼ�Ϊ+6��S��������ǿ��SO(OH)2����H2SO3���е�S��ʹ�ǻ���O��H��Ĺ��õ��ӶԸ���ƫ��Oԭ�ӣ��ǻ����������H+��������H2SO4ǿ��H2SO3 4 ![]()
����������1��Cuԭ�Ӻ�������Ų�Ϊ1s22s22p63s23p63d104s1����̬Cuԭ�ӵļ۵����Ų�ʽΪ.3d104s1 ����ȷ����.3d104s1��
2��Cuԭ�ӵļ۵���3d104s1��ʧȥ1�����Ӻ�3d10��Ϊȫ����״̬���ṹ�ȶ�������ͭԭ����ʧȥ1�����ӣ���һ�����ܽ�С������ԭ�Ӽ۵���Ϊ3d64s2��ʧȥ1�����Ӻ����ȶ��ṹ�����ԣ���ԭ�Ӳ���ʧȥ1�����ӣ���һ�����ܽϴ�����I1(Fe)>I1(Cu)����ȷ����>��
��3������֪�����еĵ�ԭ������������ԭ�Ӿ���ͬһƽ������Ϊƽ�������Σ�������������Nԭ�ӵ��ӻ�����Ϊsp2 ����ȷ����sp2��
�ڸ��ݷ��ӽṹ��֪1mol���������е�����Ϊ��������1��N-H������4��C-H������2��C-N������3��C-C������������������������Ϊ10 NA �����Ի��������γɴ�������ԭ����4��̼+1����=5����������Ϊ����ԭ����δ����ɼ��ĵ���Ϊ1�ԣ�̼̼ԭ�Ӽ�����γ������⣬����4��̼�ֱ��ṩ1�������γ����������е�����Ϊ6���������Ի��еĴ�����Ӧ��ʾΪ![]() ����ȷ�𰸣�10 NA ��
����ȷ�𰸣�10 NA �� ![]() ��
��
�����Ӽ���������е���ͣ���NH3��H2O�����о��������������ԭ�ӵĵ縺�Դ��뵪ԭ�����������H2O���Ӽ�ϴ�ˮ�ķе���ߣ�����������⻯��ķе��ɵ͵��ߵ�˳��ΪCH4��NH3��H2O����ȷ�𰸣�CH4��NH3��H2O��
��Fe2+�ṩ�չ����O2�ṩ�µ��Ӷ���ͨ����λ����������ȷ������λ����
��4��S��̬3s2 3p4��1��3s��2��3p����sp2�ӻ���SO2�Ŀռ乹��Ϊƽ����������SO2(OH)2(��H2SO4)��S�Ļ��ϼ�Ϊ+6��S��������ǿ��SO(OH)2����H2SO3���е�S��ʹ�ǻ���O��H��Ĺ��õ��ӶԸ���ƫ��Oԭ�ӣ��ǻ����������H+��������H2SO4ǿ��H2SO3 ����ȷ����ƽ����������SO2(OH)2(��H2SO4)��S�Ļ��ϼ�Ϊ+6��S��������ǿ��SO(OH)2����H2SO3���е�S��ʹ�ǻ���O��H��Ĺ��õ��ӶԸ���ƫ��Oԭ�ӣ��ǻ����������H+��������H2SO4ǿ��H2SO3 ��
��5��ʯī�ľ����ṹ��ͼ��ʾ���辧���ĵױ߳�Ϊacm����Ϊhcm������dcm����h=2d����ͼ���Կ���1��ʯī�����к���4��̼ԭ��(4=8��![]() +4��
+4��![]() +2��
+2��![]() +1)��
+1)��

����ͼ��֪:a/2=r��sin 60�㣬��a=��3r��
��g��cm-3= =
= �����d=
�����d=![]() ����ȷ����4��
����ȷ����4��![]() ��
��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�����Ŀ����������������ǹ�ҵ����Ҫ�Ļ���ԭ�ϣ�Ҳ��ʵ�����ﳣ�����Լ���
�ⶨ�к��ȣ�
д��ϡ�����ϡ����������Һ��Ӧ��ʾ�к��ȵ��Ȼ�ѧ����ʽ__________________________
���к�����ֵΪ57.3kJ/mol����
��2��ȡ50mL 0.5mol/L HCl��Һ��50mL0.55mol/L NaOH��Һ���вⶨ����ʵ����ֵС��57.3kJ/mol��ԭ������_______________������ţ���
A�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�Ӳⶨ������¶�
B����ȡ��������ʱ���Ӷ���
C���ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ���
D��ʵ��װ�ñ��¡�����Ч����
������к͵ζ���
���ⶨijNaOH��Һ�����ʵ���Ũ�ȣ�����0.1000 mol��L-1 HCl����Һ�����к͵ζ�(�÷�̪��ָʾ��)����ش��������⣺
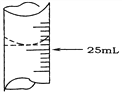
��1������ѧ����ʵ������У���¼�ζ�ǰ�ζ�����Һ�����Ϊ1.10 mL���ζ���Һ����ͼ�����ʱ���ı���Һ�����Ϊ_________________��
��2����ѧ����������ƽ��ʵ�飬���ݼ�¼���£�
ʵ����� | ����NaOH��Һ�����/mL | 0.1000mol��L-1HCl��Һ�����/mL | |
�ζ�ǰ�̶� | �ζ���̶� | ||
1 | 25.00 | 0.00 | 26.11 |
2 | 25.00 | 1.56 | 31.30 |
3 | 25.00 | 0.22 | 26.31 |
ѡȡ�����������ݣ����������NaOH��Һ�����ʵ���Ũ��Ϊ________________(С���������λ)��
��3��������Щ������ʹ�ⶨ���ƫ��___________(�����)��
A����ƿ������ˮϴ�������ô���Һ��ϴ
B����ʽ�ζ���������ˮϴ�������ñ�Һ��ϴ
C���ζ�ǰ��ʽ�ζ��ܼ������δ�ų����ζ���������ʧ
D���ζ�ǰ������ȷ���ζ����ӵζ��ܶ���
�ζ��յ���ж�__________________________