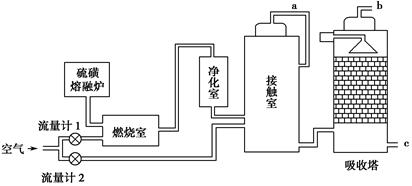
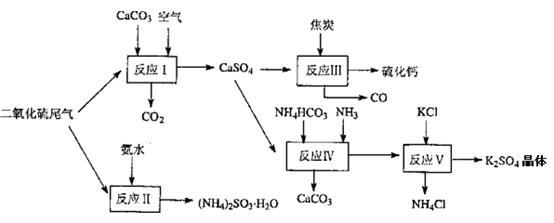
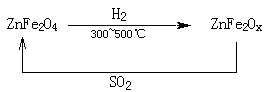
��Ŀ����
��ҵ����������ʱ�����ô�������Ӧ��SO2ת��ΪSO3��һ���ؼ����衣ѹǿ���¶ȶ�SO2ת���ʵ�Ӱ�����±�(ԭ�������ɷֵ��������Ϊ��SO2 7%��O2 11%��N2 82%)��
| ѹǿ/MPa ת����/% �¶�/�� | 0.1 | 0.5 | 1 | 10 |
| 400 | 99.2 | 99.6 | 99.7 | 99.9 |
| 500 | 93.5 | 96.9 | 97.8 | 99.3 |
| 600 | 73.7 | 85.8 | 89.5 | 96.4 |
(1)��֪SO2�������Ƿ��ȷ�Ӧ��������ñ��������ƶϴ˽��ۣ�________________________________________________________��
(2)��400��500��ʱ��SO2�Ĵ��������ó�ѹ�����Ǹ�ѹ����Ҫԭ���ǣ�__________________________________________��
(3)ѡ�����˵Ĵ������Ƿ�������SO2��ת���ʣ�________(��ǡ���)���Ƿ��������÷�Ӧ�ų���������________(��ǡ���)��
(4)Ϊ���SO3�����ʣ�ʵ����������________����SO2��
(5)��֪��2SO2(g)��O2(g)=2SO3(g) ��H����196.9 kJ��mol��1������ÿ����1���98%����������Ҫ��SO3��������SO2������ЩSO3���ų���������
(1)ѹǿһ��ʱ���¶�����ʱ��SO2ת�����½���˵�����������淴Ӧ�Ľ��У���������ӦΪ���ȷ�Ӧ
(2)����ѹǿ�����SO2ת����������Ӱ�죬���������ӳɱ�
(3)�� ��
(4)Ũ����
(5)8.0��103 t 9.845��109 kJ
����

��ϰ��ϵ�д�
�����Ŀ
���л�ѧ��ҵ�У�δ�漰ԭ��ѭ��ʹ�õ���
| A����°��Ƽ | B������ά�Ƽ |
| C����ҵ��ⱥ��ʳ��ˮ | D����ҵ���� |
���Ӳ�Ʒ�����Ĵ������������Ի��������˼������в��ij��ѧ��ȤС�齫һ����������·������õ���Cu��Al��Fe������Au��Pt�Ƚ����Ļ�������Ƴ������Ʊ�����ͭ�������������·�ߣ�
��Ҫ��ش��������⣺
(1)����1�д��ڵĽ�����_____________��
(2)��֪�������pH���±���
| | �������↑ʼ����ʱ��pH | �������������ȫʱ��pH |
| Fe2+ | 7.0 | 9.0 |
| Fe3+ | 1.9 | 3.7 |
| Cu2+ | 4.9 | 6.7 |
| Al3+ | 3.0 | 4.4 |
�����������X�������ѡ�õ���___________(����)
a�����Ը��������Һ b��Ư�� c������ d��˫��ˮ
��������Ӧ������ҺpH�ķ�Χ��________________________��
(3)������Һ2�мȲ�����Fe2+�ֲ�����Fe3+�IJ���������____________________��
(4)��һ�����ӷ���ʽ��ʾ�����Һ�м����������۵ķ�Ӧ��___________________��
(5)��CuSO4��5H2O�Ʊ�CuSO4ʱ��Ӧ��CuSO4��5H2O����______(����������)�м�����ˮ��
(6)����ijЩ������ʹ�õĸ��������һ�����Ϳɳ���أ�����ͨ���ܵ����ȣ��õ���ܳ�ʱ�䱣���ȶ��ķŵ��ѹ��������ص��ܷ�ӦΪ 3Zn + 2K2FeO4 + 8H2O
 3Zn(OH)2 + 2Fe(OH)3 + 4KOH���õ�طŵ�ʱ������ӦʽΪ_______��ÿ��1mol K2FeO4����ԭ��ת�Ƶ��ӵ����ʵ���Ϊ____�����ʱ������ӦʽΪ___________��
3Zn(OH)2 + 2Fe(OH)3 + 4KOH���õ�طŵ�ʱ������ӦʽΪ_______��ÿ��1mol K2FeO4����ԭ��ת�Ƶ��ӵ����ʵ���Ϊ____�����ʱ������ӦʽΪ___________�� ��ʶ��Ӧ�����Ի�ѧ��Ӧ��Ӱ�죬��ѧ�û�������֪ʶ������Ҫ���塣����˵������ȷ���� (����)��
| A��SO2�ϳ�SO3��Ȼ���ڷ��ȷ�Ӧ������ҵ�Ϻϳ�SO3ʱ������� |
| B���ϳɰ���ҵ���ø��¸�ѹ��ʹ�ô���������Щ���������ϻ�ѧƽ��ԭ�� |
| C���ϳɴ���Ĺ����У�Ӧ������ʳ��ˮ��ͨ��NH3����ͨ������CO2 |
| D��ͨ����ѹ���µķ����ɻ���Ʊ�NH3��ԭ����N2 |