��Ŀ����
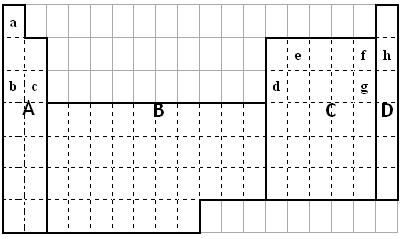
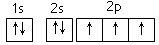
��1����Ԫ�����ڱ������ĸ������У����ʾ��ܵ������ ������
��a��A ��b��B ��c��C ��d��D
��2��������Ӳ�ֻ��Բ���ε����Ƶ�����Ԫ�������� �����С�
��a��A ��b��B ��c��C ��d��D
��3��e�γɵĵ��ʾ������Ϊ ��
| A�����Ӿ��� | B�����Ӿ��� | C��ԭ�Ӿ��� | D���������� |
���_________(�����)��
�ٱȽ����ֵ��ʵ���ɫ �ڱȽ��⻯����ȶ���
��������Ԫ�������ڱ���λ�� �ܱȽ�ԭ�Ӱ뾶��С
�ݱȽ�����������Ӧˮ���������
(5) ����һ����ѧ����ʽ��˵��b�Ľ����Ա�dǿ
_____________________________________________________________________
��1��b ��1�֣� ��2��a ��1�֣� ��3��BC (2�֣���4��1,5( 2�֣�_
��5��NaOH+Al(OH)3��NaAlO2+2H2O( 2�֣�
��5��NaOH+Al(OH)3��NaAlO2+2H2O( 2�֣�
��

��ϰ��ϵ�д�
�����Ŀ
 ������������Ӧˮ���ﷴӦ�Ļ�ѧ����ʽ
������������Ӧˮ���ﷴӦ�Ļ�ѧ����ʽ 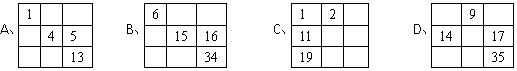 v
v �� ��
�� ��