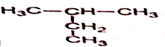��Ŀ����
2014��4��22���ǵ�45����������գ��������������Ϊ����ϧ������Դ��ת�䷢չ��ʽ����������Ϊ�����ϻ������ǣ�������
| A������̫���ܡ�ˮ�ܡ����ܡ���ȼ��������Դ������ʹ��ú��ʯ�͵Ȼ�ʯȼ�� |
| B��ʵ����Դ��������ã����迪������Դ |
| C�����²�ú�����ͼ�������߲��������㹤ҵ�����Ŀ��ٷ�չ |
| D����ʵ��Դ�ġ�3R����չ�ۣ�����������Դ���ģ�Reduce����������Դ���ظ�ʹ�ã�Reuse������Դ��ѭ��������Recycle�� |
���㣺���������������Ⱦ������
ר�⣺��ѧӦ��
��������ϧ������Դ��ת�䷢չ��ʽ���ֵ��ǽ��ܼ��ŵ�˼�룬����ˮ��Դ����ֹˮ����Ⱦ����ֹ������Ⱦ����Լ��Դ�Ĵ�ʩ�Ⱦ�������һ���⣮
���
�⣺A��̫���ܡ�ˮ�ܡ����ܡ���ȼ�����������Դ��ú��ʯ�͵Ȼ�ʯȼ��ȼ�ղ�����������Ⱦ�����̫���ܡ�ˮ�ܡ����ܡ���ȼ��������Դ������ʹ��ú��ʯ�͵Ȼ�ʯȼ�����ڻ������������Ͻ��ܼ��ŵ�ԭ��A��ȷ��
B����������Դ�������ڼ��ٻ�������Ⱦ�����Ͻ��ܼ��ŵ�ԭ��B��ȷ��
C��úΪ��������Դ��ȼ��ú����ɻ�����Ⱦ�������Ͻ��ܼ��ŵ������C����
D��������Դ���ģ�Reduce����������Դ���ظ�ʹ�ã�Reuse������Դ��ѭ��������Recycle�����Ͻ��ܼ��ŵ�ԭ��D��ȷ��
��ѡC��
B����������Դ�������ڼ��ٻ�������Ⱦ�����Ͻ��ܼ��ŵ�ԭ��B��ȷ��
C��úΪ��������Դ��ȼ��ú����ɻ�����Ⱦ�������Ͻ��ܼ��ŵ������C����
D��������Դ���ģ�Reduce����������Դ���ظ�ʹ�ã�Reuse������Դ��ѭ��������Recycle�����Ͻ��ܼ��ŵ�ԭ��D��ȷ��
��ѡC��
���������⿼������Դ�ۺ����á�������������������ɫ��ѧ��������̼�����������������˼�룬�ӻ�ѧ�ӽǷ�������������ʵ�ʣ�ѧ�����ã�

��ϰ��ϵ�д�
�����Ŀ
��ѧ����ᡢ����������������أ�����˵����ȷ���ǣ�������
| A��������ϩ������Ʒ������ʳƷ��װ |
| B�����ع��͡���ֹʳ�ã������������Ʒ��� |
| C��ú����������Һ���������仯��ת��Ϊ���ȼ�� |
| D����ά�ء���֬�������ʾ�����Ȼ�߷��ӻ����� |
����Cl-��ѡ�Լ���ȷ���ǣ�������
| A��HCl��AgNO3 |
| B��HNO3��AgNO3 |
| C��H2SO4��AgNO3 |
| D��ǰ���߶���ȷ |
�����������ʵ��ó��Ľ�����ȷ���ǣ�������
| A������NH4��2SO4��CuSO4��Һ�ֱ���뵰������Һ�������ֳ������������߾���ʹ�����ʱ��� |
| B��������ͭм��ϡ���ᷴӦֹͣ���ټ���1mol/Lϡ���ᣬͭм�����ܽ⣬������ɫ���������ͭ��ֱ����1mol/Lϡ���ᷴӦ |
| C���������³���̬��ij�л���ͨ����ˮ�У���ˮ��ɫ�������������һ����̼̼˫����̼̼���� |
| D����ij��Һ�еμ�KSCN��Һ����Һ����ɫ���μ���ˮ����Һ�Ժ�ɫ������Һ��һ����Fe2+ |
��ұ��ҵ�ϣ����õ�ⷨ�õ�K��Ca��Na��Mg��Al�Ƚ�������ԭ���ǣ�������
| A����Щ�����ܻ��� |
| B����������� |
| C����Щ�����������۵�� |
| D���ɱ��� |
����˵���У���ȷ���ǣ�������
| A��ȩ�������õ�����һԪ�� |
| B����ȩ����ȩ����ȩ��û��ͬ���칹�� |
| C����2%��ϡ��ˮ��μ���2%��AgNO3��Һ�У�������ǡ���ܽ�Ϊֹ�����Ƶ�������Һ |
| D����2%��NaOH��Һ4��6�Σ�����2mL 10%��CuSO4��Һ���Ƶ�Cu��OH��2����Һ����������ȩ�����Լ� |
����������ʵ����֪X��Y��Z��W��ԭ����ǿ������˳���ǣ�������
��X+Y2+=X2++Y
��Z+2H2O
Z��OH��2+H2��
��Z2+��X2+����������
��YԪ������������Ӧˮ����ļ��Ա�WԪ������������Ӧˮ����ļ���ǿ��
��X+Y2+=X2++Y
��Z+2H2O
| ||
��Z2+��X2+����������
��YԪ������������Ӧˮ����ļ��Ա�WԪ������������Ӧˮ����ļ���ǿ��
| A��X��Z��Y��W |
| B��Z��W��X��Y |
| C��Z��X��Y��W |
| D��Z��Y��X��W |
�����й�̫���ܵ�˵������ȷ���ǣ�������
| A��̫�����ԹⲨ��ʽ���͵����� |
| B����ʯȼ���̲ص���Դ����̫���� |
| C��̫�����ǽྻ��Դ������Ⱦ���� |
| D��ʳ��ֲ�����ڵĵ��ۡ������ʵ���ֱ��������Դ |
 ��
��
 ��
��