��Ŀ����
��08�Ϻ�12У�ڶ����������ڻ��������У�������Ʊ�ͨ�����ô���������ԭ����Ҫ�ǰ�����������
����1�����ڶ�����̼���ܱ�ˮ��ȫ���գ�������ɵ������뾭��������������յ�ѭ��������ʹ����ת��Ϊ���ᣨ������������������е�������ʧ����
��1���������Ϸ�����Ҫʹ������ȫת��Ϊ���ᣬ��ԭ�ϰ������������ʵ�����Ͷ�ϱ�����Ϊ ��
��2������������ϵ�ԭ�Ͻ�ԭ�Ϸ���һ�����з�Ӧ�����µ��ܱ������н��У��������ʲ�����罻���������������Һ����������Ϊ ��
��3��ѭ�������Ĵ�����������������������ת���ʾ���һ������ѧ��ϵ������һ�����Ķ������������ԼҪ���� ��ѭ������������ʹ����������ת����ԼΪ96%��
����2�������������Ƶõ����ᣬ����ϡ�ͳ�Ũ���ø��ֲ�ͬŨ�ȵ����ᡣ����ʵ��֤����ͬһ�����벻ͬŨ�ȵ����ᷴӦ�����ɲ�ͬ�Ļ�ԭ���ij�о�С��ʵ���о���þ�벻ͬŨ�����ᷴӦ��������������Ҫ��H2��N2��NO��NO2����Һ�еIJ�����Ҫ��Mg��NO3��2��NH4NO3��H2O��
��4������0.96Mg������Ũ��Ϊ4mol/l��HNO3��ȫ
��Ӧ���ռ���224ml��S.T.P�����壬���������
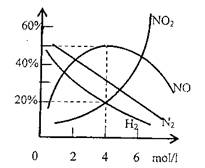
�ɷֺ���������Ũ����������ͼ��ʾ��ͨ����
��ȷ����ԭ�������ɼ����ɷֵ����ʵ���֮
���Ƕ��٣�
�𰸣���1��1��2��2�֣�
��2��77.8%��2�֣�
��3��3��2�֣�
��4��NO N2 H2 NO2 NH4+��2�֣�
5��3��1��1��4��2�֣�

 ��У����ϵ�д�
��У����ϵ�д�2(08�㽭ʡ������ѧģ��)ʵ�����и���2SO2��O2![]() 2SO3����H=-393.2 kJ?mol-1�������ͼ��ʾʵ��װ�����Ʊ�SO3���塣��ش��������⡣
2SO3����H=-393.2 kJ?mol-1�������ͼ��ʾʵ��װ�����Ʊ�SO3���塣��ش��������⡣
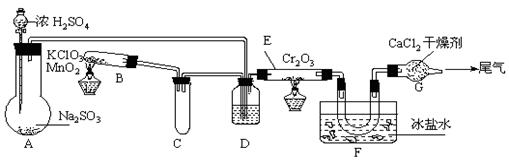 |
��1��ʵ��ǰ��������еIJ����ǣ���������ƣ�����д������̣�������������
��2����Aװ���м���Na2SO3�����ͬʱ������Ӽ���ˮ��Ȼ���ٵμ�Ũ���ᡣ�Ӽ���ˮ�������� ��
��3��С�Թ�C��������
��4�����ƿD��ʢ���Լ��� ��װ��D������������ ������ ����
�� ��
��5��ʵ���е�Cr2O3�������ʱ��Ӧ���ƾ����ƿ�һ����ټ��ȣ��Է��¶ȹ��ߣ���������ԭ���� �� ��
��6��װ��F��U�����ռ��������ʵ���ɫ��״̬��
��7��װ��G��������
��8����Gװ�õ�����β������������
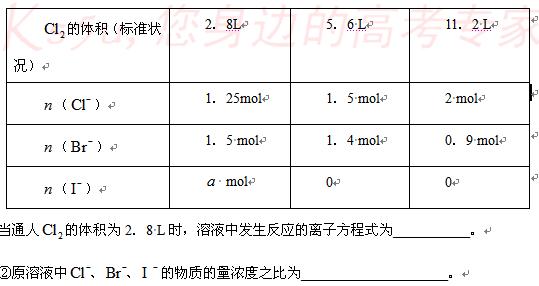
(08����8У����������)1 Lij�����Һ�����ܺ��е��������±���
���ܴ������е������� |
|
���ܴ������е������� |
|
 ��1��������Һ����μ���
��1��������Һ����μ���![]() ��Һ���ʵ�
��Һ���ʵ�
���ȣ�������������������ʵ�����![]() ��
��
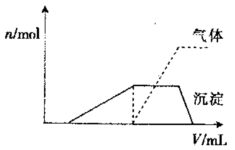
�����![]() ��Һ�������V���Ĺ�ϵ
��Һ�������V���Ĺ�ϵ
����ͼ��ʾ�������Һ��ȷ�����е�����
��_______________������ȷ���Ƿ���
����������______________��Ҫȷ�����
�ڿɲ�������ʵ����___________���϶������ڵ���������_________________��
��2������⣬����Һ�к��д�����![]() ������1 L�û����Һ��ͨ��D���ε�
������1 L�û����Һ��ͨ��D���ε�![]() ����Һ��
����Һ��![]() �����ʵ�����ͨ��
�����ʵ�����ͨ��![]() ���������״�����Ĺ�ϵ���±���ʾ��������ش��������⣺
���������״�����Ĺ�ϵ���±���ʾ��������ش��������⣺
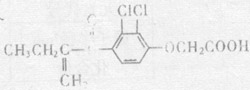 (08������Ϣ��)2008�걱�����˻����֡����İ��ˡ���һ����Ҫ�ٴ��Ǽ�������˶�Ա�����˷ܼ�����������һ�ֱ���ֹʹ�õ�ҩ���ṹ��ʽ��ͼ��ʾ�������й�������˵����ȷ���ǣ���
(08������Ϣ��)2008�걱�����˻����֡����İ��ˡ���һ����Ҫ�ٴ��Ǽ�������˶�Ա�����˷ܼ�����������һ�ֱ���ֹʹ�õ�ҩ���ṹ��ʽ��ͼ��ʾ�������й�������˵����ȷ���ǣ���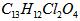
 ��Һ������ɫ��Ӧ
��Һ������ɫ��Ӧ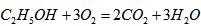 ������˵����ȷ���ǣ���
������˵����ȷ���ǣ���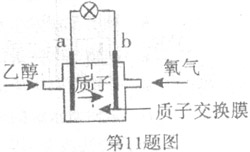 A��a��Ϊ��ص�����
A��a��Ϊ��ص�����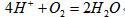
 ���صĵ���
���صĵ��� 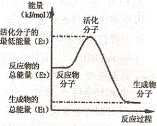 ��CH3OH��g��+H2O��g��=CO2��g��+3H2��g����H1=+49.0KJ/mol
��CH3OH��g��+H2O��g��=CO2��g��+3H2��g����H1=+49.0KJ/mol