��Ŀ����
�����������������Լ�����Ӱ���ȣ����Ʊ��������£�

(1)����(NH4)2Fe(SO4)2��6H2O��Һʱ���������ϡ���ᣬĿ����________��
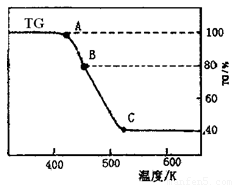
(2)���ƵõIJ�Ʒ����������н������ط������������ͼ(TG����ʾ������������ռԭ��Ʒ�������İٷ���)��

����C��ʱ������Ļ�ѧʽΪ________��
�����о�ѧ����ʵ�������������ɫ�����H2�����ղ�����Ҳ�����Ĵ����������ɣ����������һ������ʽ����������ʵ��________��
����ȡ�������146����ˮ���FeC2O41.44 g����ij��յ��ܱ������У��ٳ���0.04 mol��CO��������1100�棬����FeO(s)��CO(g)![]() Fe(s)��CO2(g)��Ӧƽ�ⳣ��K��0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣�________��
Fe(s)��CO2(g)��Ӧƽ�ⳣ��K��0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣�________��
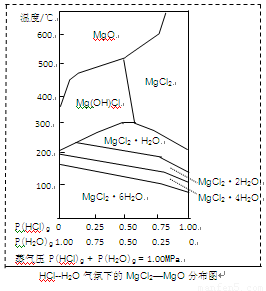
(3)����þ�ڹ�������������Ҫ���ã�����MgCl2Ϊԭ�ϻ�ȡ���¶Ⱥ�ѹǿP(HCl)g��MgCl2��6H2O�����ȷֽ�����Ӱ����ͼ��ʾ�������ͼ��ش��������⣺
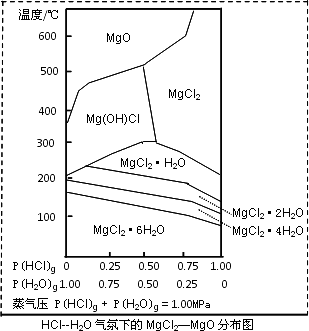
��д��P(HCl)g��0.25MPa���¶ȴ�300�����ߵ�550��ʱ��Ӧ�Ļ�ѧ����ʽ________��
��ʵ�������У���MgCl2��6H2O������ȵ�600��Ĺ����м����ò�����ˮMgCl2����ԭ����________����Ҫ�õ���ˮMgCl2���ȡ�Ĵ�ʩ��________��
������
|
����(1)����Fe2+ˮ�� ����(2)��FeO ������3FeO(s)��H2O(g) ������71.4�� ����(3)��Mg(OH)Cl ������P(HCl)С������ʱMg2+�ᷢ��ˮ�⣬��Ҫ������MgO ����ͨ��HCl���壬����P(HCl)����֤���ѹ����0.75������Mg2+ˮ�⣮ |


 6H2O��Һʱ���������ϡ���ᣬĿ����________________________________________________________��
6H2O��Һʱ���������ϡ���ᣬĿ����________________________________________________________��
 B������Ӧ�Ļ�ѧ����ʽΪ
B������Ӧ�Ļ�ѧ����ʽΪ Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣�____________________________��
Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣�____________________________��
 6H2O��Һʱ���������ϡ���ᣬĿ���� ��
6H2O��Һʱ���������ϡ���ᣬĿ���� ��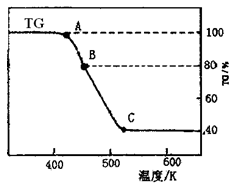
 Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣� ��
Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣� ��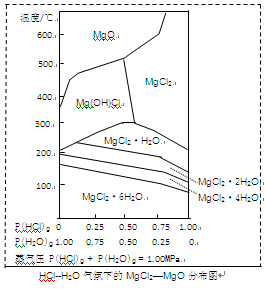

 6H2O��Һʱ���������ϡ���ᣬĿ����
��
6H2O��Һʱ���������ϡ���ᣬĿ����
��
 Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣� ��
Fe(s)+CO2(g)��Ӧƽ�ⳣ��K=0.4����÷�Ӧ��ƽ��ʱ��FeO��ת����Ϊ���٣� ��