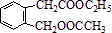��Ŀ����
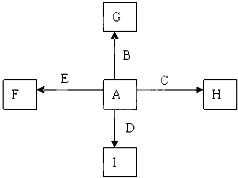
ͼ��A��B��C��D��EΪ���ʣ�G��H��I��F��B��C��D��E�ֱ��A�γɵĶ�Ԫ�������֪��
��C+G��B+H���ų��������ȣ��÷�Ӧ��Ӧ��������ĺ��ӣ�G�����Ǵ��������Ҫ�ɷ֣�
��I��һ�ֳ������������壬����E������Ӧ��2E+I��2F+D��F��EԪ�ص���������Ϊ60%��
�ش��������⣮
��1����Ӧ�ٵĻ�ѧ����ʽ�� ��
��2��������I�������ڵĻ�ѧ���� ��������ӡ����ԡ��Ǽ��ԡ�����
��3����ȡ11.9g B��C��E�Ļ����ù�����NaOH��Һ�ܽ���ˡ�����ʣ���������Ϊ9.2g����ȡ��������B��C��E�Ļ������������ϡ������ȫ�ܽ⣬���ռ������������6.72L����ʣ��Ļ��Һ�м��������NaOH��Һ�����ó���������Ϊ ��
A��27.2g B��7.8g C��2.7g D��19.4g
��4��C�������NaOH��Һ��Ӧ�����ӷ���ʽ�ǣ� ��
��5����G���ڹ�����ϡ�����У����������е�Fe3+�ķ����� ���������ѡ����ѡ����ȷѡ���ͬ�������������е�Fe2+�ķ����� ��
A���μ�KSCN��Һ����Һ��Ѫ��ɫ
B�������ۣ���Һ��dz��ɫ
C����������KMnO4��Һ��Ѹ����ɫ
D���μ�NaOH��Һ���а�ɫ������Ѹ�ٱ�ɻ���ɫ���ת��Ϊ���ɫ��
��C+G��B+H���ų��������ȣ��÷�Ӧ��Ӧ��������ĺ��ӣ�G�����Ǵ��������Ҫ�ɷ֣�
��I��һ�ֳ������������壬����E������Ӧ��2E+I��2F+D��F��EԪ�ص���������Ϊ60%��

�ش��������⣮
��1����Ӧ�ٵĻ�ѧ����ʽ��
��2��������I�������ڵĻ�ѧ����
��3����ȡ11.9g B��C��E�Ļ����ù�����NaOH��Һ�ܽ���ˡ�����ʣ���������Ϊ9.2g����ȡ��������B��C��E�Ļ������������ϡ������ȫ�ܽ⣬���ռ������������6.72L����ʣ��Ļ��Һ�м��������NaOH��Һ�����ó���������Ϊ
A��27.2g B��7.8g C��2.7g D��19.4g
��4��C�������NaOH��Һ��Ӧ�����ӷ���ʽ�ǣ�
��5����G���ڹ�����ϡ�����У����������е�Fe3+�ķ�����
A���μ�KSCN��Һ����Һ��Ѫ��ɫ
B�������ۣ���Һ��dz��ɫ
C����������KMnO4��Һ��Ѹ����ɫ
D���μ�NaOH��Һ���а�ɫ������Ѹ�ٱ�ɻ���ɫ���ת��Ϊ���ɫ��
������A��B��C��D��E�dz������ʣ��ɢٷ�ӦC+G��B+H�����ų��������ȣ�G�����Ǵ��������Ҫ�ɷ֣��÷�Ӧ����������ĺ��ӣ���CΪAl��GΪFe3O4��BΪFe��HΪAl2O3��A+C��H������AΪO2��I��һ�ֳ������������壬IΪCO2������E���Է�����Ӧ��2E+I��2F+D��F��EԪ�ص���������Ϊ60%����FΪMgO��EΪMg��DΪC��������ʵ����������
����⣺A��B��C��D��E�dz������ʣ��ɢٷ�ӦC+G��B+H�����ų��������ȣ�G�����Ǵ��������Ҫ�ɷ֣��÷�Ӧ����������ĺ��ӣ���CΪAl��GΪFe3O4��BΪFe��HΪAl2O3��A+C��H������AΪO2��I��һ�ֳ������������壬IΪCO2������E���Է�����Ӧ��2E+I��2F+D��F��EԪ�ص���������Ϊ60%����FΪMgO��EΪMg��DΪC��
��1�����з�Ӧ�Ļ�ѧ����ʽΪ8Al+3Fe3O4
9Fe+4Al2O3��
�ʴ�Ϊ��8Al+3Fe3O4
9Fe+4Al2O3��
��2��IΪCO2�����еĻ�ѧ��ΪC=O����Ϊ���Լ���
�ʴ�Ϊ�����ԣ�
��3��B��C��E�ֱ�ΪFe��Al��Mg�����������ᷴӦ���������NaOH���ֱ�����Fe��OH��3��NaAlO2��Mg��OH��2���������Ƚϣ��������ӵIJ���ΪOH-��������6.72L����ӦΪNO����ȡ11.9g B��C��E�Ļ����ù�����NaOH��Һ�ܽ���ˡ�����ʣ���������Ϊ9.2g����Al������Ϊ11.9g=9.2g=2.7g��
n��Al��=0.1mol��0.1molAlʧȥ0.3mol���ӣ�
�ɵ����غ��֪Fe��Mg��ʧȥ3��
-0.3mol=0.6mol
�ɵ����غ�͵���غ��֪Fe��OH��3��Mg��OH��2��n��OH-��=n��e-��=0.6mol��
��m��OH-��=0.6mol��17g/mol=10.2g��
���Է�Ӧ�������������Ϊ10.2g+9.2g=19.4g��
�ʴ�Ϊ��D��
��4��Al������������Ʒ�Ӧ�����ӷ���ʽΪ2Al+2OH-+2H2O�T2AlO2-+3H2����
�ʴ�Ϊ��2Al+2OH-+2H2O�T2AlO2-+3H2����
��5��GΪFe3O4�������ᷴӦ����Fe3+��Fe2+������Fe3+���ɵμ�KSCN��Һ����Һ��Ѫ��ɫ������Fe2+���ɵ�������KMnO4��Һ��Ѹ����ɫ��
�ʴ�Ϊ��A��C��
��1�����з�Ӧ�Ļ�ѧ����ʽΪ8Al+3Fe3O4
| ||
�ʴ�Ϊ��8Al+3Fe3O4
| ||
��2��IΪCO2�����еĻ�ѧ��ΪC=O����Ϊ���Լ���
�ʴ�Ϊ�����ԣ�
��3��B��C��E�ֱ�ΪFe��Al��Mg�����������ᷴӦ���������NaOH���ֱ�����Fe��OH��3��NaAlO2��Mg��OH��2���������Ƚϣ��������ӵIJ���ΪOH-��������6.72L����ӦΪNO����ȡ11.9g B��C��E�Ļ����ù�����NaOH��Һ�ܽ���ˡ�����ʣ���������Ϊ9.2g����Al������Ϊ11.9g=9.2g=2.7g��
n��Al��=0.1mol��0.1molAlʧȥ0.3mol���ӣ�
�ɵ����غ��֪Fe��Mg��ʧȥ3��
| 6.72L |
| 22.4L/mol |
�ɵ����غ�͵���غ��֪Fe��OH��3��Mg��OH��2��n��OH-��=n��e-��=0.6mol��
��m��OH-��=0.6mol��17g/mol=10.2g��
���Է�Ӧ�������������Ϊ10.2g+9.2g=19.4g��
�ʴ�Ϊ��D��
��4��Al������������Ʒ�Ӧ�����ӷ���ʽΪ2Al+2OH-+2H2O�T2AlO2-+3H2����
�ʴ�Ϊ��2Al+2OH-+2H2O�T2AlO2-+3H2����
��5��GΪFe3O4�������ᷴӦ����Fe3+��Fe2+������Fe3+���ɵμ�KSCN��Һ����Һ��Ѫ��ɫ������Fe2+���ɵ�������KMnO4��Һ��Ѹ����ɫ��
�ʴ�Ϊ��A��C��
���������⿼��������ƶϣ�������ѧ���ķ�������������������ʵ�������Ŀ��飬Ϊ�߿��������ͣ��Ѷ��еȣ�ע��������ʵ����ʡ���;��������Ϊ������Ĺؼ���

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
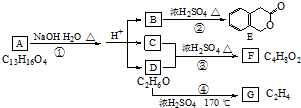
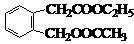 +2NaOH
+2NaOH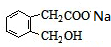 +CH3COONa+CH3CH2OH
+CH3COONa+CH3CH2OH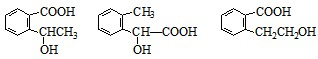 ������һ��
������һ�� ����CH2=CH2+H2O
����CH2=CH2+H2O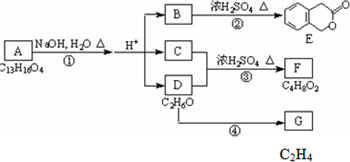 ������ͼ�ش����⣺
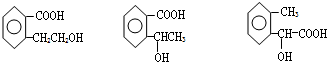
������ͼ�ش����⣺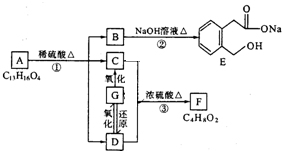 ͼ��A��B��C��D��E��F��G��Ϊ�л������B������ֻ��һ����״�ṹ������ͼʾ�ش����⣺
ͼ��A��B��C��D��E��F��G��Ϊ�л������B������ֻ��һ����״�ṹ������ͼʾ�ش����⣺