��Ŀ����
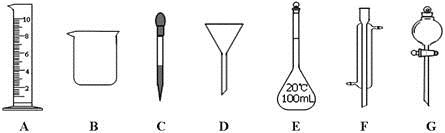
(1)д���������ƣ� F ��
(2)����ʵ��������õ�����G���� (ѡ������ѡ��ı����ĸ)��
a������ˮ��CC14�Ļ���� b������ˮ�;ƾ��Ļ���� c������ˮ����ɰ�Ļ����
(3)ʵ��������210mL 0.5mol/L��������Һ��
�����й�������E��ʹ�÷����У���ȷ���� (ѡ������ѡ��ı����ĸ)��
a��ʹ��ǰӦ����Ƿ�©Һ b��ʹ��ǰ������
c�������������ʷ�Ӧ���ܽ������ d������Һ��ֱ��ת�Ƶ�����ƿ��
������ȡ10mol/L��Ũ���� mL��ȡ�ø��������ʱ����Ҫ�õ����������е�A�� (ѡ�������ı����ĸ)��
��֪25��ʱ����������ʵĵ���ƽ�ⳣ���������±���
��ѧʽ | CH3COOH | H2CO3 | HClO | |
ƽ�ⳣ�� | Ka=1.8��10-5 | Ka1=4.3��10-7 | Ka2=5.6��10-11 | Ka=3.0��10-8 |
�ش��������⣺
(1) ���ʵ���Ũ�Ⱦ�Ϊ0��1mol��Lһ1��������Һ��a��CH3COONa b��Na2CO3 c��NaClO d��NaHCO3��pH��С���������˳����___________ (����ĸ��ʾ)
(2) �����£�0��1mol��L��1��CH3COOH��Һ��ˮϡ�����У����б���ʽ�����ݱ�����_______ (�����)
A��c��H+�� B��c(H+)��c(CH3COOH)
C��c(H+)��c(OH��) D��c(OH-)��c(H+)

(3) �����Ϊl00mL pH=2��CH3COOH��һԪ��HX����ˮϡ������pH����Һ����Ĺ�ϵ��ͼ��ʾ����HX�ĵ���ƽ�ⳣ��_______ (���������������=��)CH3COOH�ĵ���ƽ�ⳣ����
(4) 25��ʱ����CH3COOH��CH3COONa�Ļ����Һ�У������pH=6������Һ�У�
�� c(CH3COO��)��c(Na+)=_______ mol��L��1(�ȷֵ)
�� c(CH3COO-)��c(CH3COOH)=_______ ��


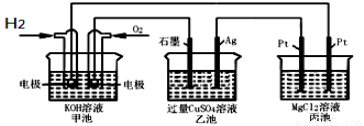
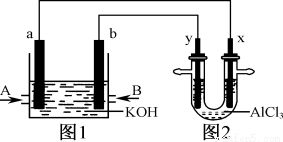
 Cl2����H2����2OH��
Cl2����H2����2OH��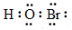

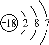
 Na����H����CO
Na����H����CO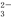
 ��˵���в���ȷ����
��˵���в���ȷ����