��Ŀ����
�±����г���25�桢101kPaʱһЩ���ʵ�ȼ��������
��֪���ܣ�C-H����413.4kJ/mol��H-H����436.0kJ/mol��������������ȷ���ǣ�������
| ���� | CH4 | C2H2 | H2 |
| ȼ����/kJ/mol | 890.3 | 1299.6 | 285.8 |
| A��C��C����Ϊ796.0 kJ/mol | ||
| B��2CH4��g��+2O2��g���T2H2O��l����H=-890.3 kJ/mol | ||
C��H2��g��+
| ||
| D��2CH4��g���TC2H2��g��+3H2��g����H=-376.4 kJ/mol |
���㣺��Ӧ�Ⱥ��ʱ�
ר�⣺��ѧ��Ӧ�е������仯
������A�����ݻ�ѧ��Ӧ���ʱ�=��Ӧ���ܼ���-��������ܼ��ܣ�
B��ȼ������1mol��ȼ��ȼ�շų���������
C��ȼ������ָ��ȼ��ȼ�������ȶ��Ļ�����ʱ�ų���������H����Һ̬ˮ��
D��2CH4��g���TC2H2��g��+3H2��g��Ϊ���ȷ�Ӧ����H��0��
B��ȼ������1mol��ȼ��ȼ�շų���������
C��ȼ������ָ��ȼ��ȼ�������ȶ��Ļ�����ʱ�ų���������H����Һ̬ˮ��
D��2CH4��g���TC2H2��g��+3H2��g��Ϊ���ȷ�Ӧ����H��0��
���
�⣺A��CH4��g��+2O2��g��=CO2��g��+2H2O��l����H3=-890.3kJ/mol�٣�C2H2��g��+
O2��g���T2CO2��g��+H2O��l����H=-1299.6 kJ/mol�ڣ�H2��g��+
O2��g��=H2O��l����H=-285.8kJ/mol�ۣ����ݢ�+�ۡ�3-�١�2�ɵã�C2H2��g��+3H2��g���T2CH4��g����H=-376.4 kJ/mol����H=��Ӧ���ܼ���-�������ܼ���=C��C���ļ���+2��413.4kJ/mol+3��436.0kJ/mol-8��413.4kJ/mol=-376.4KJ/mol����C��C���ļ���=796.0 kJ/mol����A��ȷ��
B��ȼ������1mol��ȼ��ȼ�շų���������B�С�H=-890.3��2 kJ/mol����B����
C������ȼ���ȱ���������Һ̬ˮ�Ƿų�����������C����
D����A��֪��C2H2��g��+3H2��g���T2CH4��g����H=-376.4 kJ/mol���� 2CH4��g���TC2H2��g��+3H2��g����H=+376.4 kJ?mol-1����D����
��ѡ��A��
| 5 |
| 2 |
| 1 |
| 2 |
B��ȼ������1mol��ȼ��ȼ�շų���������B�С�H=-890.3��2 kJ/mol����B����
C������ȼ���ȱ���������Һ̬ˮ�Ƿų�����������C����
D����A��֪��C2H2��g��+3H2��g���T2CH4��g����H=-376.4 kJ/mol���� 2CH4��g���TC2H2��g��+3H2��g����H=+376.4 kJ?mol-1����D����
��ѡ��A��
������������Ҫ������ȼ���ȵ�Ӧ�ã�����ȼ���ȵĶ����˹���ɵ������ǽ����Ĺؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
����ԭ�ӻ�ԭ�����У������ڹ����ŵ��ǣ�������
| A����ԭ�� | B���ǻ� | C������ | D������ |
ij�绯ѧװ����ͼ������˵����ȷ���ǣ�������
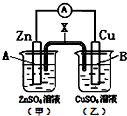

| A����X�����ţ��������B�缫�����·����A�缫 |
| B����X��ͭ������A�缫�ĵ缫��Ӧʽ�ǣ�Zn2++2e-�TZn |
| C����X��ʯī�������ҳ���Һ��pH���С |
| D����X��п������A�缫���������С |
��֪25��ʱ�й�����ĵ���ƽ�ⳣ����
������˵����ȷ���ǣ�������
| ���ữѧʽ | CH3COOH | HCN | H2CO3 |
| ����ƽ�ⳣ����25�棩 | 1.8��10-5 | 4.9��10-10 | Ka1=4.3��10-7 Ka2=5.6��10-11 |
| A�������ʵ���Ũ�ȵĸ���Һ pH ��ϵΪ��pH�� NaCN����pH��Na2CO3����pH�� CH3COONa�� |
| B��Ũ�Ⱦ�Ϊ0.2mol/L NaHCO3�� Na2CO3��Һ�У����ڵ����������ͬ |
| C��0.4mol?L-1 HCN ��Һ�� 0.2mol?L-1 NaOH ��Һ�������Ϻ�������Һ�У�c��Na+����c��OH-����c��H+�� |
| D��25��ʱ��pH=4��Ũ�Ⱦ�Ϊ0.1 mol?L-1��CH3COOH��CH3COONa�����Һ�У�c��Na+��+c��H+����c��CH3COO-��+c��CH3COOH�� |
 ����������ѧ���㣬����̼�ṹ�����֣�����һ�ֵ���̼Ӳ�ȳ������ʯ���壬��Ϊ��һָ�ij�Ӳ�²��ϣ���֪�õ���̼�Ķ�ά����ṹ��ͼ��ʾ�������йص���̼��˵����ȷ���ǣ�������
����������ѧ���㣬����̼�ṹ�����֣�����һ�ֵ���̼Ӳ�ȳ������ʯ���壬��Ϊ��һָ�ij�Ӳ�²��ϣ���֪�õ���̼�Ķ�ά����ṹ��ͼ��ʾ�������йص���̼��˵����ȷ���ǣ�������| A������̼���ڷ��Ӿ��� |
| B������̼�ķ���ʽΪC3N4 |
| C���þ����е�̼����ԭ�Ӻ��ⶼ����8�����ȶ��ṹ |
| D��ÿ��̼ԭ�����ĸ���ԭ��������ÿ����ԭ��������̼ԭ������ |
���л�ѧ����ʽ�������ڹ�ҵ��ұ������þ���ǣ�������
A��2Na+MgCl2
| ||||
B��MgCl2 ���ۻ���
| ||||
C��2MgO
| ||||
D��MgO+C
|
��Ԫ�����ڱ��Ľ���Ԫ�غͷǽ���Ԫ�ؽ��紦���������ҵ��IJ����ǣ�������
| A���ư뵼��IJ��� |
| B�����¡���ʴ�ĺϽ���� |
| C����ũҩ�IJ��� |
| D���ƴ����IJ��� |