��Ŀ����
ʵ������NaOH��������250mL 1��25mol/L��NaOH��Һ����ղ���ش��������⣺
��1������ʱ����IJ��������У��ձ����������� �� ��
��2������ʱ������ȷ�IJ���˳���ǣ���ĸ��ʾ��ÿ����ĸֻ����һ�Σ� ��
A����30mLˮϴ���ձ�2��3�Σ�ϴ��Һ��ע������ƿ����
B������ƽȷ��ȡ�����NaOH����������������ˮ��Լ30mL�����ò���������������ʹ�����ܽ�
C��������ȴ��NaOH��Һ�ز�����ע��250mL������ƿ��
D��������ƿ�ǽ����ߵ�ҡ��
E�����ý�ͷ�ιܼ�ˮ��ʹ��Һ����ǡ����̶�����
F������������ƿ��С�ļ�ˮ��ֱ��Һ��ӽ��̶�1��2cm��
��3���������Ƶ���ҺŨ��ƫ�͵��� ��
A������NaOHʱ���������������
B��������ƿ��ת����Һʱ(ʵ�鲽��C)������Һ����������ƿ����
C��������ˮʱ���������˿̶���
D������ʱ���ӿ̶���
E������ǰ������ƿ������������ˮ
��4��ijͬѧ���ù���Na2CO3����Na2CO3��Һ�Ĺ�����ͼ��ʾ���������������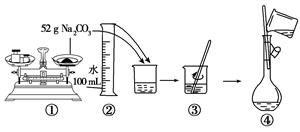
��1������ʱ����IJ��������У��ձ����������� �� ��
��2������ʱ������ȷ�IJ���˳���ǣ���ĸ��ʾ��ÿ����ĸֻ����һ�Σ� ��
A����30mLˮϴ���ձ�2��3�Σ�ϴ��Һ��ע������ƿ����
B������ƽȷ��ȡ�����NaOH����������������ˮ��Լ30mL�����ò���������������ʹ�����ܽ�
C��������ȴ��NaOH��Һ�ز�����ע��250mL������ƿ��
D��������ƿ�ǽ����ߵ�ҡ��
E�����ý�ͷ�ιܼ�ˮ��ʹ��Һ����ǡ����̶�����
F������������ƿ��С�ļ�ˮ��ֱ��Һ��ӽ��̶�1��2cm��
��3���������Ƶ���ҺŨ��ƫ�͵��� ��
A������NaOHʱ���������������
B��������ƿ��ת����Һʱ(ʵ�鲽��C)������Һ����������ƿ����
C��������ˮʱ���������˿̶���
D������ʱ���ӿ̶���
E������ǰ������ƿ������������ˮ
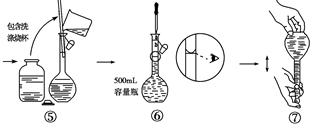
��4��ijͬѧ���ù���Na2CO3����Na2CO3��Һ�Ĺ�����ͼ��ʾ���������������


��1�� 250 ml ����ƿ ��ͷ�ι� ��2�� BCAFED ��3�� ABC ��4�� �٢�
�����������1������ʵ������IJ����Լ�ÿ��������Ҫ����ȷ����Ӧ��������������250mL��Һ����ѡ������ƿ�Ĺ��Ϊ250mL������ʱ�����м��㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȵȲ�����һ����������ƽ��������ҩ��ȡ��ҩƷ��NaOHӦ�����ձ��г��������ձ����ܽⲢ�ò��������裬��ȴ��ת�Ƶ�250mL����ƿ�У����ò���������������ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�������Ҫ������Ϊ��ƽ��ҩ�ס��ձ���250mL����ƿ������������ͷ�ιܣ������������ձ���250mL����ƿ������������ͷ�ιܡ�
��2������ʵ�鲽���������֪��˳��ΪBCAFED
��3���������������ʵ����ʵ��������Һ�������Ӱ�죬����c=n/V������A������NaOHʱ��������������̣����ʵ��������٣�Ũ��ƫ�ͣ���ȷ��B��������ƿ��ת����Һʱ������Һ����������ƿ���棬���ʵ��������٣�Ũ��ƫ�ͣ���ȷ��C��������ˮʱ���������˿̶��ߣ���Һ���ƫ��Ũ��ƫ�ͣ���ȷ��D������ʱ���ӿ̶��ߣ���Һ���ƫС��Ũ��ƫ�ߣ�����E������ǰ������ƿ������������ˮ����Ӱ�죬Ũ�Ȳ��䣬����
��4���ٳ���Na2CO3ʱ��������������̣�����ʱ���ӿ̶��ߡ�

��ϰ��ϵ�д�
 ȫ�ܲ��һ���þ�ϵ�д�
ȫ�ܲ��һ���þ�ϵ�д�
�����Ŀ
 ��SO42-��Fe3+��H+��M�������ʵ���֮��Ϊ��n��NO
��SO42-��Fe3+��H+��M�������ʵ���֮��Ϊ��n��NO ���Un��M����2�U3�U1�U3�U1����M�����������е�
���Un��M����2�U3�U1�U3�U1����M�����������е�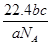 L
L