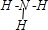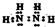��Ŀ����
��2013?�Ͽ���һģ����ͼ��ʾΪ���������Ʊ������롢�����������֤�IJ�������װ�ã������豸���г̶ֹ�װ�þ���ȥ���������Ҫ��ش��������⣨����װ�ÿ�����ѡ�ã���Ҫʱ���ظ�ѡ��a��bΪ��������

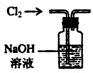
��1�����������ͨ��CO��CO2�Ļ�����壬E�ڷ���CuO��ѡ��װ�û�ô��������CO������֤�仹ԭ�Լ����������ѡװ�õ�����˳��Ϊ
��2��ֹͣCO��CO2��������ͨ�룬E�ڷ���Na2O2����A��E��D��B��Hװ��˳����ȡ���������O2������O2�����Ҵ�����ʱ������aӦ
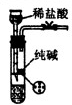
��3����������ڸ�ͨ��������Һ©���ڸļ�Ũ��ˮ��Բ����ƿ�ڸļ�NaOH���壬E�ڷ��ò���Ͻ�������A��G��E��Dװ��˳����ȡ����İ���������֤����ijЩ���ʣ�
��װ��A���ܲ���������ԭ����
��ʵ���й۲쵽E���к���ɫ������֣�֤����������
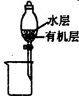
��4��ͼ1��ʾΪʵ������ȡ�����ķ���װ�ã�����װ�������ԵIJ�����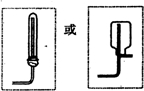
 ��
��

��1�����������ͨ��CO��CO2�Ļ�����壬E�ڷ���CuO��ѡ��װ�û�ô��������CO������֤�仹ԭ�Լ����������ѡװ�õ�����˳��Ϊ
ACBECF
ACBECF
������ĸ���ţ�������֤CO���������������AB֮���Cװ������Һ���ֳ��壬EF֮���Cװ������Һ�����
AB֮���Cװ������Һ���ֳ��壬EF֮���Cװ������Һ�����
����2��ֹͣCO��CO2��������ͨ�룬E�ڷ���Na2O2����A��E��D��B��Hװ��˳����ȡ���������O2������O2�����Ҵ�����ʱ������aӦ
�ر�
�ر�
������bӦ��
��
����Ҫ���ȵ�����װ����k��m
k��m
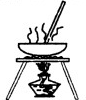
������ĸ���ţ���m�з�Ӧ�Ļ�ѧ����ʽΪ��2CH3CH2OH+O2
2CH3CHO+2H2O
| Cu |
| �� |
2CH3CH2OH+O2
2CH3CHO+2H2O
��| Cu |
| �� |
��3����������ڸ�ͨ��������Һ©���ڸļ�Ũ��ˮ��Բ����ƿ�ڸļ�NaOH���壬E�ڷ��ò���Ͻ�������A��G��E��Dװ��˳����ȡ����İ���������֤����ijЩ���ʣ�
��װ��A���ܲ���������ԭ����
������������ˮ�ų������ȣ��¶����ߣ�ʹ�����ܽ�ȼ�С���ų�������������ˮ����ʹ���ų����������Ƶ������OH-������OH-Ũ�ȣ���ʹ��ˮ����ƽ�����ƣ����°����ų�
������������ˮ�ų������ȣ��¶����ߣ�ʹ�����ܽ�ȼ�С���ų�������������ˮ����ʹ���ų����������Ƶ������OH-������OH-Ũ�ȣ���ʹ��ˮ����ƽ�����ƣ����°����ų�
����ʵ���й۲쵽E���к���ɫ������֣�֤����������
��ԭ
��ԭ
�ԣ���4��ͼ1��ʾΪʵ������ȡ�����ķ���װ�ã�����װ�������ԵIJ�����
���ӵ��ܣ������ܲ���ˮ�У������Թܵ����ܿ������ݲ�����ֹͣ���ȣ���������ˮ�������γ�һ���ȶ�����ˮ��
���ӵ��ܣ������ܲ���ˮ�У������Թܵ����ܿ������ݲ�����ֹͣ���ȣ���������ˮ�������γ�һ���ȶ�����ˮ��
���������߿��ڻ����ռ�������װ��ͼ

��������1����NaOH��Һ����ʯ��ˮ��ȥCO2����Ũ�������CO����CuO����CO���ó���ʯ��ˮ����CO���������ȼ�շ���ȥ�����CO��
��2��ֹͣCO��CO2��������ͨ�룬aӦ�رգ�b��Ũ������NaOH��Һ��Ӧ�ų��������ӿ�A�е�ˮ��������H2O��E��Na2O2��Ӧ������O2������k������CH3CH2OH�Ļӷ�������m��CH3CH2OH��O2��Ӧ����CH3CHO��
��3��������ƽ���ƶ��Լ���������Һ�е��ܽ�ȷ�����
��NH3��O2������֤���������л�ԭ�ԣ�
��4��װ�������Լ����ԭ���ǣ�ͨ�����巢�����븽���Һ�幹�ɷ����ϵ�����ݸı���ϵ��ѹǿʱ���������������ݵ����ɡ�ˮ�����γɡ�Һ��������ȣ����ж�װ�������Եĺû��������ȿ����Ἣ������ˮ����Ҫ�������������ռ���
��2��ֹͣCO��CO2��������ͨ�룬aӦ�رգ�b��Ũ������NaOH��Һ��Ӧ�ų��������ӿ�A�е�ˮ��������H2O��E��Na2O2��Ӧ������O2������k������CH3CH2OH�Ļӷ�������m��CH3CH2OH��O2��Ӧ����CH3CHO��
��3��������ƽ���ƶ��Լ���������Һ�е��ܽ�ȷ�����
��NH3��O2������֤���������л�ԭ�ԣ�
��4��װ�������Լ����ԭ���ǣ�ͨ�����巢�����븽���Һ�幹�ɷ����ϵ�����ݸı���ϵ��ѹǿʱ���������������ݵ����ɡ�ˮ�����γɡ�Һ��������ȣ����ж�װ�������Եĺû��������ȿ����Ἣ������ˮ����Ҫ�������������ռ���
����⣺��1��Ҫ��ô��������CO�ͱ�����A�е�NaOH��Һ����CO2����ͨ��C�еij���ʯ��ˮ�����֤��CO2�ѱ���ȫ���գ���ͨ��BŨ�������CO���壮COͨ��E�м��ȵ�CuO��������CO2����C�г���ʯ��ˮ���ձ���ǣ�֤��CO��ԭ�Լ����������ѡװ�õ�����˳��ΪACBECF��
�ʴ�Ϊ��ACBECF��AB֮���Cװ������Һ���ֳ��壬EF֮���Cװ������Һ����ǣ�
��2��ֹͣCO��CO2��������ͨ���Ҫ�رջ���a������b����ϡH2SO4��NaOH �����кͷ�Ӧ��������ˮ������Aװ���г�������E��Na2O2��Ӧ�ͻ���O2���ɣ�������NaOH����D�У�O2��B��ŨH2SO4�����ٽ���Hװ�ý��Ҵ�������O2���ؼ��ȵ�ͭ˿������������ȩ����Ӧ�Ļ�ѧ����ʽΪ2CH3CH2OH+O2
2CH3CHO+2H2O��
�ʴ�Ϊ���رգ���k��m��2CH3CH2OH+O2
2CH3CHO+2H2O��
��3��������ƽ���ƶ��Լ���������Һ�е��ܽ�ȷ�����������������ˮ�ų������ȣ��¶����ߣ�ʹ�����ܽ�ȼ�С���ų�������������ˮ����ʹ���ų����������Ƶ������OH-�����˰�ˮ��OH-Ũ�ȣ���ʹ��ˮ����ƽ�����ƣ����°����ų���
�ʴ�Ϊ��������������ˮ�ų������ȣ��¶����ߣ�ʹ�����ܽ�ȼ�С���ų�������������ˮ����ʹ���ų����������Ƶ������OH-������OH-Ũ�ȣ���ʹ��ˮ����ƽ�����ƣ����°����ų���
�ڰ���ͨ��G�м�ʯ�ұ������E�в���Ͻ�Ĵ���������O2��Ӧ����������NO��NO�Ӵ�O2�ͻᱻ�����ɺ���ɫ��NO2���壮NH3��O2������֤���������л�ԭ�ԣ��ʴ�Ϊ����ԭ��
��4���Ʊ�����ǰ����װ�õ�������Ϊ�������ܵ�һ�˷���ˮ�У������Թ�һ����������ܿ�������������ð��ʱ��ֹͣ���ȣ���������ˮ�������ڵ��ܿ��γ�һ����Һ����ȶ�ˮ�����������Ժã��ռ������������������ռ���װ��Ϊ ��
��
�ʴ�Ϊ�������ܵ�һ�˷���ˮ�У������Թ�һ����������ܿ�������������ð��ʱ��ֹͣ���ȣ���������ˮ�������ڵ��ܿ��γ�һ����Һ����ȶ�ˮ�����������Ժã� ��
��
�ʴ�Ϊ��ACBECF��AB֮���Cװ������Һ���ֳ��壬EF֮���Cװ������Һ����ǣ�
��2��ֹͣCO��CO2��������ͨ���Ҫ�رջ���a������b����ϡH2SO4��NaOH �����кͷ�Ӧ��������ˮ������Aװ���г�������E��Na2O2��Ӧ�ͻ���O2���ɣ�������NaOH����D�У�O2��B��ŨH2SO4�����ٽ���Hװ�ý��Ҵ�������O2���ؼ��ȵ�ͭ˿������������ȩ����Ӧ�Ļ�ѧ����ʽΪ2CH3CH2OH+O2
| Cu |
| �� |
�ʴ�Ϊ���رգ���k��m��2CH3CH2OH+O2
| Cu |
| �� |
��3��������ƽ���ƶ��Լ���������Һ�е��ܽ�ȷ�����������������ˮ�ų������ȣ��¶����ߣ�ʹ�����ܽ�ȼ�С���ų�������������ˮ����ʹ���ų����������Ƶ������OH-�����˰�ˮ��OH-Ũ�ȣ���ʹ��ˮ����ƽ�����ƣ����°����ų���
�ʴ�Ϊ��������������ˮ�ų������ȣ��¶����ߣ�ʹ�����ܽ�ȼ�С���ų�������������ˮ����ʹ���ų����������Ƶ������OH-������OH-Ũ�ȣ���ʹ��ˮ����ƽ�����ƣ����°����ų���
�ڰ���ͨ��G�м�ʯ�ұ������E�в���Ͻ�Ĵ���������O2��Ӧ����������NO��NO�Ӵ�O2�ͻᱻ�����ɺ���ɫ��NO2���壮NH3��O2������֤���������л�ԭ�ԣ��ʴ�Ϊ����ԭ��
��4���Ʊ�����ǰ����װ�õ�������Ϊ�������ܵ�һ�˷���ˮ�У������Թ�һ����������ܿ�������������ð��ʱ��ֹͣ���ȣ���������ˮ�������ڵ��ܿ��γ�һ����Һ����ȶ�ˮ�����������Ժã��ռ������������������ռ���װ��Ϊ
 ��
���ʴ�Ϊ�������ܵ�һ�˷���ˮ�У������Թ�һ����������ܿ�������������ð��ʱ��ֹͣ���ȣ���������ˮ�������ڵ��ܿ��γ�һ����Һ����ȶ�ˮ�����������Ժã�
 ��
�����������⿼����������Ʊ����ռ������롢�����������֤�������Լ�飬�������˽̲��еĶ�����֪ʶ���ݣ���Na2O2�����ʣ��Ҵ�������������Ӧ�����Ĵ�������Ӧ�ȣ�����˵����������൱�ḻ������һ��ʵ���������ڶ���ʵ�����ڸ߿�����Ҳ�dz��ټ���

��ϰ��ϵ�д�
 ���Ͱ�ͨ��ĩ���ϵ�д�
���Ͱ�ͨ��ĩ���ϵ�д�
�����Ŀ