��Ŀ����
15�������������壨FeSO4•7H2O����ҽҩ������Ѫ����ij����С��ⶨ�ò�Ѫ������Ԫ�صĺ�����ʵ�鲽�����£�
��1������ڼ������H2O2��Ŀ���ǽ�Fe2+ȫ������ΪFe3+��
��2��������з�Ӧ�����ӷ���ʽΪFe3++3NH3•H2O=Fe��OH��3��+3NH4+��
��3���������һϵ�д����IJ������裺���ˡ�ϴ�ӡ����ա���ȴ��������
��4��ʵ������Ũ��������250mL1mol/L��ϡ���ᣬ����ʱ�õ��IJ������������ձ����⣬����250mL����ƿ����Ͳ������������ͷ�ιܣ�
��5����ʵ������ģ���ÿƬ��Ѫ������Ԫ�ص�����0.07ag���ú�a�Ĵ���ʽ��ʾ����
���� ������ͼ��֪����ʵ��ԭ��Ϊ����ҩƷ�е�Fe2+�γ���Һ����Fe2+����ΪFe3+��ʹFe3+ת��Ϊ����������������ת��Ϊ��������ͨ���ⶨ�����������������㲹Ѫ������Ԫ�صĺ�����
��1��˫��ˮ���������ԣ������������ܽ�Fe2+ȫ������ΪFe3+��������ͼ��֪������H2O2�ǽ�Fe2+����ΪFe3+��
��2��������ǽ�Fe3+ת��Ϊ��������������
��3���������һϵ�д�������������������Һ����ת��Ϊ����������Ҫ���ˡ�ϴ�ӵ�����������Ȼ��������������������ȴ�������������������
��4������һ�������1mol/L��ϡ���ᣬ����ʱ��Ҫ����������Ͳ���������ձ�����ͷ�ιܣ�����ƿ��
��5��������Ԫ���غ��֪ag����������Ԫ�ص�������Ϊ10Ƭ��Ѫ���������������Դ������㣮
��� �⣺������ͼ��֪����ʵ��ԭ��Ϊ����ҩƷ�е�Fe2+�γ���Һ����Fe2+����ΪFe3+��ʹFe3+ת��Ϊ����������������ת��Ϊ��������ͨ���ⶨ�����������������㲹Ѫ������Ԫ�صĺ�����
��1��˫��ˮ���������ԣ������������ܽ�Fe2+ȫ������ΪFe3+���ʴ�Ϊ����Fe2+ȫ������ΪFe3+��
��2��������ǽ�Fe3+ת��Ϊ����������������Ӧ���ӷ���ʽΪFe3++3NH3•H2O=Fe��OH��3��+3NH4+���ʴ�Ϊ��Fe3++3NH3•H2O=Fe��OH��3��+3NH4+��
��3���������һϵ�д�������������������Һ����ת��Ϊ����������Ҫ���ˡ�ϴ�ӵ�����������Ȼ��������������������ȴ�������������������
�ʴ�Ϊ�����ˣ����գ�
��4������һ�������1mol/L��ϡ���ᣬ����ʱ��Ҫ���������ձ���250mL����ƿ����Ͳ������������ͷ�ιܣ��ʴ�Ϊ��250mL����ƿ����Ͳ������������ͷ�ιܣ�
��5��ag����������Ԫ�ص�������Ϊ10Ƭ��Ѫ������������������ÿƬ��Ѫ������Ԫ�ص�����Ϊ$\frac{ag��\frac{112}{160}}{10}$=0.07ag��
�ʴ�Ϊ��0.07a��
���� ���⿼�����ʺ����IJⶨ��Ϊ��Ƶ���㣬���������еķ�Ӧ�������ⶨ���غ㷨Ӧ��Ϊ���Ĺؼ������ط�������������Ŀ��飬��Ŀ�ѶȲ���

 �����ҵ���������ϵ�д�
�����ҵ���������ϵ�д�| ѡ�� | X | Y | Z | M |
| A | Fe��s�� | FeCl2 | FeCl3 | Cl2 |
| B | NaOH��aq�� | Na2CO3 | NaHCO3 | CO2 |
| C | NH3��g�� | NO | NO2 | O2 |
| D | S��s�� | SO2 | SO3 | O2 |
| A�� | A�� | B�� | B�� | C�� | C�� | D�� | D�� |
| A�� | ��Ӧ����ֻ��O2���ɣ���6.72 L�����壨����� | |
| B�� | ������һ���漰��������ѧ��Ӧ | |
| C�� | ���յõ����������ij��� | |
| D�� | ���յõ�����Һ��c��NaCl��=0.15 mol•L-1 |
| A�� | 2gH2���е���ԭ����ĿΪNA | |
| B�� | �����£�1molCl2������NaOH��Һ��ȫ��Ӧ��ת�Ƶĵ�����Ϊ2NA | |
| C�� | ���³�ѹ�£�11.2L�������е�ԭ����ĿΪNA | |
| D�� | 1L1 mol•L-1 KOH��Һ�к��еļ�������ĿΪNA |
| A�� | 19%��MgCl2 ��Һ�У�Cl-��H2O������Ϊ4��45 | |
| B�� | Ϊ����������Һ��Cl-�� SO42-���ȼ���������Һ����ȥ��������Һ�м����ᱵ��Һ | |
| C�� | S8��NO2���ǹ��ۻ����NH4Cl ��CaC2�������ӻ����� | |
| D�� | ����AijԪ�ص�ԭ������Ϊm����ͬ���ڢ�AԪ�ص�ԭ����������Ϊm+10 |
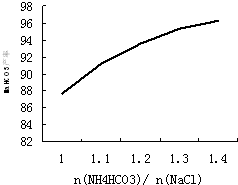 ̼�������һ����Ҫ����Σ�ʵ�����У���������̼ͨ�백ˮ���Ƶ�̼����泥��ھ��Ʊ���ʳ��ˮ�м���̼����刺��Ʊ�С�մ�NaHCO3��������ȡ�Ȼ����Ϊ���ϻ��һ���ᴿΪ��ҵ�Ȼ�泥����������գ�
̼�������һ����Ҫ����Σ�ʵ�����У���������̼ͨ�백ˮ���Ƶ�̼����泥��ھ��Ʊ���ʳ��ˮ�м���̼����刺��Ʊ�С�մ�NaHCO3��������ȡ�Ȼ����Ϊ���ϻ��һ���ᴿΪ��ҵ�Ȼ�泥����������գ�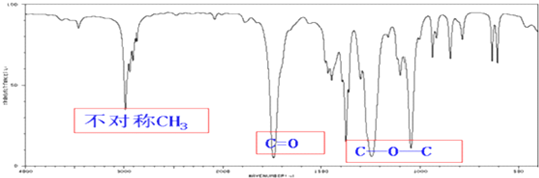
 ij�¶��£�2L�����ܱ������У�X��Y��Z�������巢����ѧ��Ӧʱ�����ʵ�����ʱ��仯�Ĺ�ϵ������ͼ��ʾ����
ij�¶��£�2L�����ܱ������У�X��Y��Z�������巢����ѧ��Ӧʱ�����ʵ�����ʱ��仯�Ĺ�ϵ������ͼ��ʾ����