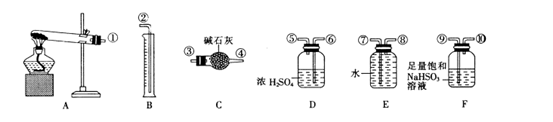
��Ŀ����
̼���ơ���������Ӻ���(aNa2CO3��bH2O2)����Ư�ס�ɱ�����á�ʵ�����á����������Ʊ������ʵ�ʵ�鲽�����£�
��1����ȡ����̼�����ܽ���һ����ˮ�������ƿ�У��ټ��������ȶ���(MgCl2��Na2SiO3)��������ȡ�
��2����������30%��H2O2��Һ�ڽ���״̬�µ�����ƿ�У���15 �����ҷ�Ӧ1 h��
��3������Ӧ��Ϻ��ټ���������ˮ�Ҵ������á��ᾧ�����ˡ�����ò�Ʒ��
(1)��1���У��ȶ�����ˮ��Ӧ�������ֳ�����������仯ѧ����ʽΪ___________________________________________________________��
(2)��2���У���Ӧ����Ϊ15 �����ҿɲ�ȡ�Ĵ�ʩ��_____________________
___________________________________________________��
(3)��3���У���ˮ�Ҵ���������____________________________________��
(4)H2O2�ĺ����ɺ�����Ʒ�����ӡ��ֳ�ȡm g(Լ0.5 g)��Ʒ��������й�������ˮ���Ƴ�250 mL��Һ��ȡ25.0 mL����ƿ�У�����ϡ�����ữ������c mol��L��1 KMnO4��Һ�ζ����յ㡣
������250 mL��Һ����IJ����������ձ�������������Ͳ________��________��
�ڵζ��յ�۲쵽��������______________________________________��
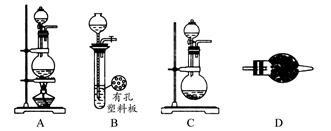
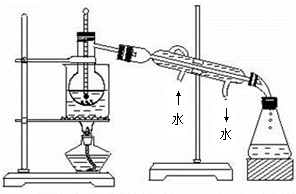
(5)��ģ�������ⶨ��Ʒ��̼���Ƶĺ�����װ������ͼ��ʾ(���Ⱥ̶�װ������ȥ)��ʵ�鲽�����£�
����1������ͼ��ʾ��װ���������װ�������ԡ�
����2��ȷ��ȡ(4)��������Һ50 mL����ƿ�С�
����3��ȷ��ȡ40.00 mLԼ0.2 mol��L��1 NaOH��Һ���ݣ��ֱ�ע���ձ�����ƿ�С�
����4������K1��K2���رջ���K3����ͨ�뵪��һ��ʱ��ر�K1��K2����K3������Һ©������ƿ�м���10 mL 3 mol��L��1������Һ��
����5����������ƿ�е�Һ����ڣ���������һ��ʱ�䡣
����6����K1�ٻ���ͨ�뵪��һ��ʱ�䡣
����7������ƿ�м������ָʾ������c1 mol��L��1 H2SO4����Һ�ζ����յ㣬����H2SO4����ҺV1 mL��
����8����ʵ�鲽��1��7�ظ����Ρ�
�ٲ���3�У�ȷ��ȡ40.00 mL NaOH��Һ����Ҫʹ�õ�������________��
�ڲ���1��7�У�ȷ�����ɵĶ�����̼������������Һ��ȫ���յ�ʵ�鲽����________(�����)��
��Ϊ�����Ʒ��̼���Ƶĺ��������貹���ʵ����______________________��
��(1)MgCl2��Na2SiO3��2H2O===2NaCl��Mg(OH)2����H2SiO3����(2)15 ��ˮԡ����ˮԡ��(3)����̼���Ƶ��ܽ��(�����ھ�������)
(4)��250 mL����ƿ����ͷ�ιܡ�����Һ�ʷۺ�ɫ��30 s����ɫ
(5)�ټ�ʽ�ζ��ܡ���1,5,6������H2SO4����Һ�ζ�NaOH��Һ��Ũ��
����

 ȫ�ſ��䵥Ԫ�����������ܸ�ϰϵ�д�
ȫ�ſ��䵥Ԫ�����������ܸ�ϰϵ�д� Ʒѧ˫�ž�ϵ�д�
Ʒѧ˫�ž�ϵ�д�����ͭ���ȷֽ���������ͭ�����壬�����¶Ȳ�ͬ������ɷ�Ҳ��ͬ������ɷֿ��ܺ�SO2��SO3��O2�е�һ�֡����ֻ����֡�ij��ѧ����С��ͨ�����̽����ʵ�飬�ⶨ��Ӧ������SO2��SO3��O2�����ʵ�����������ȷ�������ʵĻ�ѧ���������Ӷ�ȷ��CuSO4�ֽ�Ļ�ѧ����ʽ��ʵ���õ�����������ͼ��ʾ��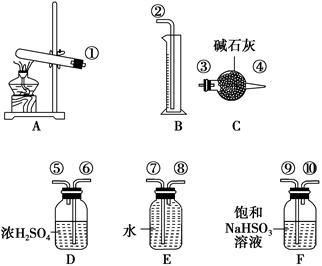
[�������]
��.��������ijɷֿ���ֻ��SO3һ�֣�
��.��������ijɷֿ��ܺ���________���֣�
��.��������ijɷֿ��ܺ���________���֡�
[ʵ��̽��]
ʵ����������ԡ���֪ʵ�����ʱ������ͭ��ȫ�ֽ⡣
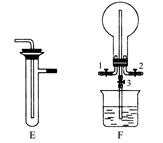
��1��������װ̽��ʵ���װ�ã����������ҵķ��������ӿڵ�����˳��Ϊ�١��������ޡ��ݡ�________��________��________��________���ڣ���ӿ���ţ���
��2����ʵ�����ʱB����Ͳû���ռ���ˮ����֤������________��ȷ��
��3��������ʵ��С����и�ʵ�飬���ڼ���ʱ���¶Ȳ�ͬ��ʵ����������������Ҳ��ͬ���������£�
| ʵ�� С�� | ��ȡCuSO4 ������/g | װ��C���� ������/g | ��Ͳ��ˮ���������ɱ�״������������/mL |
| һ | 6.4 | 2.56 | 448 |
| �� | 6.4 | 2.56 | 224 |
��ͨ�����㣬�ƶϳ���һС��͵ڶ�С���ʵ��������CuSO4�ֽ�Ļ�ѧ����ʽ��
��һС�飺________________________________________________________��
�ڶ�С�飺________________________________________________________��
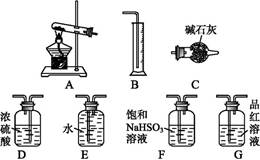
ij�ִ����к�����ɳ��Ca2����Mg2����Fe3����SO�����ʡ�ijͬѧ��ʵ����������������ִ����Ʊ����εķ�������(���ڳ������Լ��Թ���)��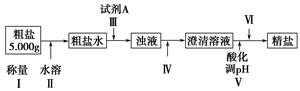
��ش��������⣺
(1)Ϊ������ѡ����������(�ñ����ĸ��д)��________��
| A���ձ� | B���Թ� | C�������� | D����Һ©����E��©����F���ƾ��� |
(2)�������г���Na2CO3��Һ��NaOH��Һ��BaCl2��Һ��Ϊ�����Լ������������Լ���˳��Ϊ��NaOH��Һ��________��________��
(3)�������У��жϼ���BaCl2�ѹ����ķ�����_________________________��
(4)������Ӧѡ�������________����������������������Ⱥ�˳��Ե��������ʵ����������Ӱ����__________________________________________��
(5)��������________(ѡ��������������ƣ��ñ����ĸ�������Ⱥ�˳����д)��
a�����ˡ�ϴ�� b��������Ũ�� c����ȡ����Һ d����ȴ���ᾧ
����(H2C2O4)��һ�����ᣬ�ڹ�ҵ������Ҫ���á�ijͬѧ�����ϵ�֪�������и��������Բ����κ�̼���Ρ�������������ĥ��֭����ˮ���ݣ������˵õ���Һ������������CaCl2��Һ��������ɫ���������ˡ�ϴ�ӳ������ã�Ȼ������ɵij�������̽����
(1)��������Һ�������Ե�ԭ����_____________________________________��
(2)��ͬѧ�����ɵij������ж���̽����
������������衣
����1��ֻ����CaCO3��
����2���ȴ���CaCO3��Ҳ����CaC2O4��
����3��___________________________________________________________��
�ڻ��ڼ���2�����ʵ�鷽��������ʵ�顣�����±���д��ʵ�鲽���Լ�Ԥ������ͽ��ۡ���ѡʵ���Լ���
1 mol��L��1 H2SO4��0.1 mol��L��1���ᡢ0.01 mol��L��1 KMnO4��Һ������ʯ��ˮ��
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡ�����������Թ��У�����_________________________________ ________________________________ | _______________________________ _______________________________ ˵����������CaCO3 |
| ����2��_________________________ ________________________________ | ________________________________ |
(3)���ڼ���3����һλͬѧ�����ɵij������ж���̽������ϡ�����ܽ����ɵij���������ˮ���Ƴ�100 mL��Һ��ÿ��ȷ��ȡ25.00 mL����Һ����0.010 mol��L��1 KMnO4����Һ�ζ���ƽ�����ı���ҺV mL����������Ʒ������Ϊm g�����в��ἰ������(��C2O42����)����������Ϊ________��
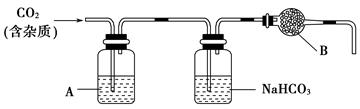
��Ԫ�ض��˵Ľ����dz���Ҫ�������ǴӺ������з�������ʵ⣨I2��������ͼ��
��1�����̢ٵ�ʵ�����������____________��____________��
��2�����������У�����������ԭ��Ӧ����_______�����ӷ���ʽΪ ��
��3�����̢۵�ʵ�����������___________��___________���ڴ˹����У��ɹ�ѡ�����
���ܼ���_______������ĸ���ţ���
| A���ƾ� | B��CCl4 | C���� | D������ |
�� ��
�� ��
�� ��
�� �����м����������������������۷֡���