��Ŀ����
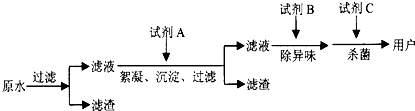
����ˮ�����Ҫ��Ⱦ��֮һ�����Ե���ˮ��������������ѧʵ���У���ʹij���е��к�������Ϊ��һ���ķ�Ӧ��γ�һ��ѭ�����Ϳɲ������ŷŸ��к����ʡ����紦������ʵ��ѭ��ʾ��ͼ����

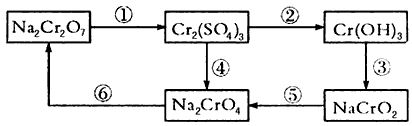
(1)����ͼ��ʾ�ı�Ų����У����з�Ӧ�����õ��Լ��������ữ��H2O2����д���÷�Ӧ�����ӷ���ʽ
_____________________
(2)���к�Cr3+��Fe3+����Һ��ʹ��NaOH��Һ��������Һ���ɽ���������������룬������ͼ��Ϣ���������漰�Ĺ��˲����У������������ӵĴ�����̬�ֱ���____________��д��ѧʽ����
(3)���Ļ��ϼ۳���ʾ��ͼ���漰��+3��+6���⣬����0��+2��+4��+5�۵ȡ�����24 mLŨ��Ϊ0.05mol/L��Na2SO3��Һǡ����20 mLŨ��Ϊ 0. 02 mol/L��Na2Cr2O7��Һ��ȫ��Ӧ����֪Na2SO3��Na2Cr2O7����Ϊ
Na2SO4����Ԫ��Cr�ڻ�ԭ�����еĻ��ϼ�Ϊ___________��
(4)��֪298 Kʱ��Cr(OH)3���ܶȻ�����Ksp=6.3��10-31(mol/L)4��ȡ����Cr2(SO4)3��Һ������һ�����İ�ˮ�ﵽ�����ܽ�ƽ�⣬���pH=11������¶��²�������Һ��Cr3+��Ũ��Ϊ__________mol/L��
_____________________
(2)���к�Cr3+��Fe3+����Һ��ʹ��NaOH��Һ��������Һ���ɽ���������������룬������ͼ��Ϣ���������漰�Ĺ��˲����У������������ӵĴ�����̬�ֱ���____________��д��ѧʽ����
(3)���Ļ��ϼ۳���ʾ��ͼ���漰��+3��+6���⣬����0��+2��+4��+5�۵ȡ�����24 mLŨ��Ϊ0.05mol/L��Na2SO3��Һǡ����20 mLŨ��Ϊ 0. 02 mol/L��Na2Cr2O7��Һ��ȫ��Ӧ����֪Na2SO3��Na2Cr2O7����Ϊ
Na2SO4����Ԫ��Cr�ڻ�ԭ�����еĻ��ϼ�Ϊ___________��
(4)��֪298 Kʱ��Cr(OH)3���ܶȻ�����Ksp=6.3��10-31(mol/L)4��ȡ����Cr2(SO4)3��Һ������һ�����İ�ˮ�ﵽ�����ܽ�ƽ�⣬���pH=11������¶��²�������Һ��Cr3+��Ũ��Ϊ__________mol/L��
(1)Cr2O72-+8H++3H2O2=2Cr3++3O2��+7H2O
(2)CrO2-��Fe(OH)3
(3)+3
(4)6.3��10-22
(2)CrO2-��Fe(OH)3
(3)+3
(4)6.3��10-22

��ϰ��ϵ�д�
 �����Ծ���ĩ���100��ϵ�д�
�����Ծ���ĩ���100��ϵ�д� ˫��ͬ������ѵ��ϵ�д�
˫��ͬ������ѵ��ϵ�д�
�����Ŀ