��Ŀ����
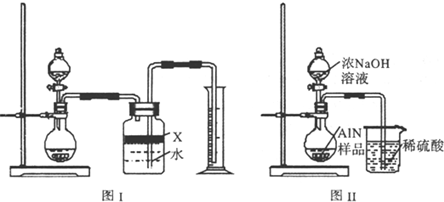
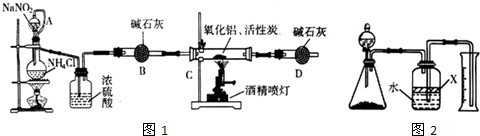
��������AIN����һ�����������ϣ��㷺Ӧ���ڼ��ɵ�·��������ij��ѧ�о�С������ ��ȡ�������������ͼ1��ʾ��ʵ��װ�ã�
�Իش�
��1��ʵ�����ñ���NaNO2�� NH4C��Һ��ȡ�����Ļ�ѧ����ʽΪ ��
��2��װ���з�Һ©����������ƿ֮��ĵ���A�������� ����д��ţ���
a����ֹNaNO2 ������Һ���� b����֤ʵ��װ�ò�©�� c��ʹNaNO2 ������Һ������
��3����ͼ���Ӻ�ʵ��װ�ã����װ�������Եķ����� ��
��4����ѧ�о�С���װ�ô����������⣬��˵���Ľ��İ취 ��
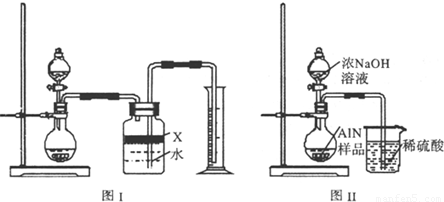
��5����Ӧ������ijͬѧ��ͼ2��ʾװ�ý���ʵ�����ⶨ�������� Ʒ������������ʵ���е���������Բ��ƣ�����֪����������NaOH��Һ��Ӧ����Na[Al��OH��4]�Ͱ�����
�ٹ��ƿ�е��Լ�X���ѡ�� ����д��ţ���a������ b���ƾ� c��ֲ���� d��CCl4
�ڹ��ƿ�е�Һ��û��װ�����Ϸ����пռ䣩����ʵ����NH3������� ���ƫ����ƫС���������䡱����
����ʵ���г�ȡ��������Ʒ������Ϊ10.0g����ð��������Ϊ3.36L����״����������Ʒ��AlN����������Ϊ ��

�Իش�
��1��ʵ�����ñ���NaNO2�� NH4C��Һ��ȡ�����Ļ�ѧ����ʽΪ
��2��װ���з�Һ©����������ƿ֮��ĵ���A��������
a����ֹNaNO2 ������Һ���� b����֤ʵ��װ�ò�©�� c��ʹNaNO2 ������Һ������
��3����ͼ���Ӻ�ʵ��װ�ã����װ�������Եķ�����
��4����ѧ�о�С���װ�ô����������⣬��˵���Ľ��İ취
��5����Ӧ������ijͬѧ��ͼ2��ʾװ�ý���ʵ�����ⶨ�������� Ʒ������������ʵ���е���������Բ��ƣ�����֪����������NaOH��Һ��Ӧ����Na[Al��OH��4]�Ͱ�����
�ٹ��ƿ�е��Լ�X���ѡ��
�ڹ��ƿ�е�Һ��û��װ�����Ϸ����пռ䣩����ʵ����NH3�������
����ʵ���г�ȡ��������Ʒ������Ϊ10.0g����ð��������Ϊ3.36L����״����������Ʒ��AlN����������Ϊ
��������1������NaNO2�� NH4Cl��Һ��Ӧ�����Ȼ��ơ�������ˮ��
��2������ʵ�����Ҫ��ϴ���ѹǿԭ�����ش�
��3��ֻҪ�Ƚ�װ���ܷ���������������ԭ��������������֤��
��4��ʵ�������ж��������β����������ֹ������Ⱦ��
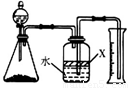
��5���ٸ��ݹ��ƿ�е��Լ�X�����ÿ��ǣ�
�ڸ��ݷ�Ӧǰ����ƿ���Ϸ����е������ռ��������仯���ǣ�
�۸��ݰ����������������ʵ������ó����е�ԭ�ӵ����ʵ��������ݵ�ԭ���غ㣬�����㵪�����İٷֺ�����
��2������ʵ�����Ҫ��ϴ���ѹǿԭ�����ش�
��3��ֻҪ�Ƚ�װ���ܷ���������������ԭ��������������֤��
��4��ʵ�������ж��������β����������ֹ������Ⱦ��
��5���ٸ��ݹ��ƿ�е��Լ�X�����ÿ��ǣ�
�ڸ��ݷ�Ӧǰ����ƿ���Ϸ����е������ռ��������仯���ǣ�
�۸��ݰ����������������ʵ������ó����е�ԭ�ӵ����ʵ��������ݵ�ԭ���غ㣬�����㵪�����İٷֺ�����
����⣺��1������NaNO2�� NH4Cl��Һ��Ӧ�����Ȼ��ơ�������ˮ���ʴ�Ϊ��NaNO2+NH4Cl
NaCl+N2��+2H2O��
��2��װ���з�Һ©����������ƿ֮��ĵ���A����ƽ����ѹ�����ã��������Ա�֤NaNO2������Һ�����£���ѡc��
��3���رշ�Һ©�����أ�ʹװ�ô����ܱ���ϵ��������һ�˽���ˮ�У����ֽ�����ƿ��ڣ�����������������ƿ�������������ͣ�������ܿ�������ð����˵�����������ã�����װ��©�����ʴ�Ϊ���ڸ����Dĩ������һ���ܣ������ܲ����ձ��е�Һ���£��þƾ�����������ƿ�����ܿ�������ð���������ƾ���һ��ʱ�䣬����������һ��ˮ����֤�����������ã�
��4��ʵ�������ж��������β����������ֹ������Ⱦ���ʴ�Ϊ���ڸ����Dĩ������һ��β������װ�ã�
��5���ٹ��ƿ�е��Լ�X�������ǽ�������ˮ��������Ϊ������������ˮ�����ھƾ��������������ˮ���ܣ����Բ����þƾ���Ҫ��ֲ���ͽ�������ˮ������ˮ�������ͣ���ѡc��
�ڷ�Ӧǰ���ƿ���Ϸ����е������ռ������ǿ�������Ӧ����ƿ���Ϸ����е������ռ������ǰ��������������˿�ʼ�Ŀ����������ռ��г���İ������ɿ�ʼʱ�Ŀ������ɣ����Զ�ʵ����û��Ӱ�죬�ʴ�Ϊ�����䣻
��5���������к���̼�����������ʣ�������������NaOH��Һ����ʵ����������۲쵽��ƿ�л��й��壬˵�����岻��������������Һ������������̼��
��6�����������Ϊ3.36L����״���������ʵ�����0.15mol�����Ե����̵����ʵ�����0.15mol��������0.15mol��41g/mol=6.15g�����Ե���������������Ϊ
��100%=61.5%���ʴ�Ϊ��61.5%��
| ||
��2��װ���з�Һ©����������ƿ֮��ĵ���A����ƽ����ѹ�����ã��������Ա�֤NaNO2������Һ�����£���ѡc��
��3���رշ�Һ©�����أ�ʹװ�ô����ܱ���ϵ��������һ�˽���ˮ�У����ֽ�����ƿ��ڣ�����������������ƿ�������������ͣ�������ܿ�������ð����˵�����������ã�����װ��©�����ʴ�Ϊ���ڸ����Dĩ������һ���ܣ������ܲ����ձ��е�Һ���£��þƾ�����������ƿ�����ܿ�������ð���������ƾ���һ��ʱ�䣬����������һ��ˮ����֤�����������ã�
��4��ʵ�������ж��������β����������ֹ������Ⱦ���ʴ�Ϊ���ڸ����Dĩ������һ��β������װ�ã�
��5���ٹ��ƿ�е��Լ�X�������ǽ�������ˮ��������Ϊ������������ˮ�����ھƾ��������������ˮ���ܣ����Բ����þƾ���Ҫ��ֲ���ͽ�������ˮ������ˮ�������ͣ���ѡc��
�ڷ�Ӧǰ���ƿ���Ϸ����е������ռ������ǿ�������Ӧ����ƿ���Ϸ����е������ռ������ǰ��������������˿�ʼ�Ŀ����������ռ��г���İ������ɿ�ʼʱ�Ŀ������ɣ����Զ�ʵ����û��Ӱ�죬�ʴ�Ϊ�����䣻
��5���������к���̼�����������ʣ�������������NaOH��Һ����ʵ����������۲쵽��ƿ�л��й��壬˵�����岻��������������Һ������������̼��
��6�����������Ϊ3.36L����״���������ʵ�����0.15mol�����Ե����̵����ʵ�����0.15mol��������0.15mol��41g/mol=6.15g�����Ե���������������Ϊ
| 6.15 |
| 10 |
�����������Ҫ֪����ȡ�����һ�㲽�裬֪������ˮ���ⶨ��������ķ��������ڰ�����������ˮ��Ҫ��ֲ���ͽ�������ˮ�ֿ����ۺ���ǿ���Ѷȴ�

��ϰ��ϵ�д�
 �»����ܶ�Ա��ϵ�д�
�»����ܶ�Ա��ϵ�д� ����ͼ����ּ��������ҵ֣�ݴ�ѧ������ϵ�д�
����ͼ����ּ��������ҵ֣�ݴ�ѧ������ϵ�д�
�����Ŀ


 ��
��