��Ŀ����
I��20mL0.1mol/LBaCl2��Һ��Cl-�����ʵ���Ũ��Ϊ________��Ba2+�����ʵ���Ϊ_______��
II����5.1gþ���Ͻ�ķ�ĩ��������������У��õ�5.6 LH2����״���£���
��1���Ͻ���þ�����ʵ���_________��
��2��д���úϽ���������NaOH��Һ�Ļ�ѧ����ʽ_________��ͬʱ����H2���������״���£�Ϊ____________ ��
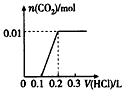
III����һ��������AlCl3��MgCl2�����Һ������һ����������������NaOH��Һ�����ɳ��������ʵ��������NaOH�����ʵ����Ĺ�ϵ��ͼ��ʾ����
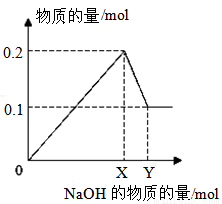
������Mg(OH)2������Ϊ__________��
��X=_______��Y=_________��
 ˫��ͬ������ѵ��ϵ�д�
˫��ͬ������ѵ��ϵ�д� �Ƹ�С״Ԫͬ������������ϵ�д�
�Ƹ�С״Ԫͬ������������ϵ�д�����ʵ�顰�����������롰���ۡ���Ӧ��ϵ��ȷ���ǣ� ��
ѡ�� | ʵ����������� | ʵ����� |
A | ��ij��Һ�м����Ȼ�����Һ���а�ɫ�������ɣ��ټ������ữ���������ܽ� | ����Һ��һ������SO42�� |
B | ��ij��Һ��ͬʱ���뼸��KSCN��Һ���������Ƶ���ˮ����Һ��Ϊ��ɫ | ����Һ��һ������Fe2+ |
C | ��ij��Һ�м���ϡ���ᣬ������ʹ����ʯ��ˮ����ǵ���ɫ���� | ����Һ��һ������CO32�� |
D | ��ij��Һ����NaOH���ȣ������ܹ�ʹʪ��ĺ�ɫʯ����ֽ��������ɫ���� | ����Һ��һ������NH4+ |
A. A B. B C. C D. D



 B.
B. 
 D.
D. 

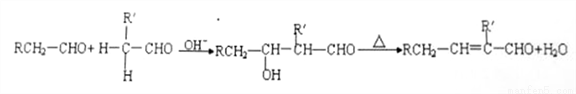

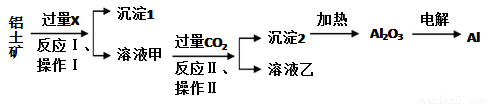
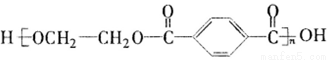
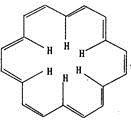
 F�Ļ�ѧ����ʽΪ___________��
F�Ļ�ѧ����ʽΪ___________��