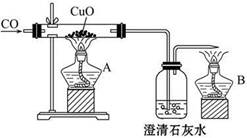
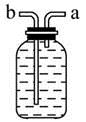
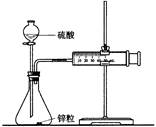
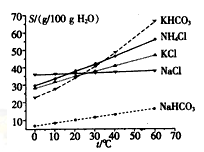
��Ŀ����
�����±��ṩ��������ҩƷ�����ܴﵽ��Ӧʵ��Ŀ�ĵ���
| ��� | ���� | ҩƷ | ʵ��Ŀ�� |
| A | ������ƽ�������룩��250mL����ƿ����Ͳ���ձ���ҩ�ס������� | NaOH���塢����ˮ | ����250mLһ�����ʵ���Ũ�ȵ�NaOH��Һ |
| B | ��Һ©������ƿ�����ܼ���Ƥ�� | ϡ���ᡢ̼���ơ���������Һ | ֤���ǽ����ԣ�S��C��Si |
| C | ��ʽ�ζ��ܡ���ʽ�ζ��ܡ���ͷ�ιܡ�����̨�������У�����ƿ | ��֪Ũ�ȵ�NaOH��Һ���������ᡢ����ˮ����ֽ | �ⶨϡ��������ʵ���Ũ�� |
| D | ����̨�������У����ƾ��ơ����Թܡ�����ƿ�����ܼ���Ƥ�� | �Ȼ�� | ��ȡ���� |
B
���������A.����250mLһ�����ʵ���Ũ�ȵ�NaOH��Һ,��Ҫʹ�õ�������������ƽ�������룩��250mL����ƿ����Ͳ���ձ���ҩ�ס�����������ͷ�ιܡ�����B��Ԫ�صķǽ�����Խǿ��������������Ӧ��ˮ��������Ծ�Խǿ�����ݸ��ֽⷴӦ�Ĺ��ɣ�ǿ����������η�Ӧ��ȡ���ᡣ��ȷ��C.�ⶨϡ��������ʵ���Ũ��,������ҩƷ��ȱ�ٴ���HCl��Һ�����ָʾ��������D.��ʵ������ȡ����Ҫ���Ȼ�����������ƹ��壬ȱ��ҩƷ�������ơ�����

��ϰ��ϵ�д�
 ���ſ����ϵ�д�
���ſ����ϵ�д�
�����Ŀ

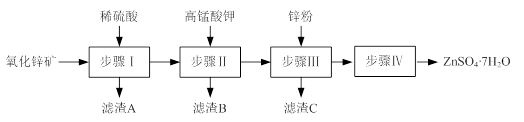
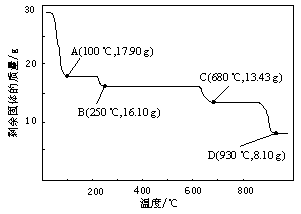
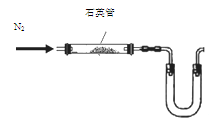
 ��
��