��Ŀ����
�ס��ҡ�����X��Ϊ��ѧ�����Ĵ��������֮��������ת����ϵ(����������ȥ)
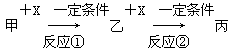
�Իش��������⣺
(1)��X��ǿ�����Ե��ʣ��������__________��
a��S��b��N2��c��Na��d��Mg��e��Al
(2)��X�ǽ������ʣ������ˮ��Һ�еμ�AgNO3��Һ������������ϡHNO3�İ�ɫ���������ҵĻ�ѧʽΪ________________������Һ������ʱӦ��������X��������(�ñ�Ҫ�����ֺ����ӷ���ʽ��ʾ)__________________________��
(3)���ס��ҡ���Ϊ���н���Ԫ�ص��������XΪǿ����ʣ�����Һ�����Һ��Ӧ�����ң���Ӧ�ٵ����ӷ���ʽΪ____________________����____________________��
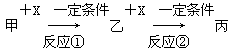
�Իش��������⣺
(1)��X��ǿ�����Ե��ʣ��������__________��
a��S��b��N2��c��Na��d��Mg��e��Al
(2)��X�ǽ������ʣ������ˮ��Һ�еμ�AgNO3��Һ������������ϡHNO3�İ�ɫ���������ҵĻ�ѧʽΪ________________������Һ������ʱӦ��������X��������(�ñ�Ҫ�����ֺ����ӷ���ʽ��ʾ)__________________________��
(3)���ס��ҡ���Ϊ���н���Ԫ�ص��������XΪǿ����ʣ�����Һ�����Һ��Ӧ�����ң���Ӧ�ٵ����ӷ���ʽΪ____________________����____________________��
(1)abc �� (2)FeCl3��2Fe3���� Fe=3Fe2������ֹFe2����������
(3)Al3����3OH����Al(OH)3����AlO2-��H����H2O=Al(OH)3��
(3)Al3����3OH����Al(OH)3����AlO2-��H����H2O=Al(OH)3��
(1)��X��ǿ��������˵������Ԫ�ؿɱ�ۻ���������ۣ���Mg��Alû�б�ۡ�������a��b��c��(2)����Һ����AgNO3��Һ������������ϡHNO3�İ�ɫ������˵������Ԫ�أ���Ϊ��������ΪFeCl3��XΪ������ΪFeCl2��Һ��Ϊ��ֹ�������������(3)�ס��ҡ����Ǻ��н���Ԫ�ص����XΪǿ����ʣ�����Һ�����Һ��������ң��������Ļ���������ʣ����Է�Ӧ�ٿ�дΪAl3����3OH��=Al(OH)3����AlO2-��H����H2O=Al(OH)3����

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
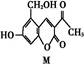
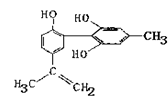
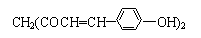 �������Ƴɵ��Լ����Լ������ʵ�����ԡ��ܹ���1 mol�û�������Ӧ��Br2(ˮ��Һ)��H2����������ֱ���
�������Ƴɵ��Լ����Լ������ʵ�����ԡ��ܹ���1 mol�û�������Ӧ��Br2(ˮ��Һ)��H2����������ֱ��� M(���ۻ�����)
M(���ۻ�����)