��Ŀ����
��1����7�֣����������л��������Żش����и��ʣ�
�� 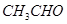 ��
�� 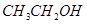 ��
��
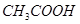 ��
�� 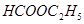
������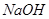 ��Һ��Ӧ����_________________��
��Һ��Ӧ����_________________��
������������Һ��Ӧ������������____________________��
�ۼ��ܷ�����ȥ��Ӧ�����ܷ���������Ӧ����___________________��
�ܺ˴Ź�������ͼ����3�����շ壬�����Ϊ3��2��1����___________��
��2������8�֣�Ϊ���Т����еij���ʵ�飨������Ϊ���ʣ�����Ӣ�����ѡ��һ���Լ����Ӣ�����ѡ��һ���ʵ�ʵ������������룬��������ں���Ĵ����ڡ�
|
�� ����ʵ�� |
�� �Լ� |
�� �������� |
�� |
||
|
�� |
�� |
�� |
|||
|
�ٱ������ӣ� |
A������Na2CO3��Һ B��NaCl���� C��NaOH��Һ D��CaO |
a������ b����Һ c������������ d����ȡ |
�� |
|
|
|
���Ҵ���ˮ�� |
�� |
|
|
||
|
�۷��������͡�ˮ�� |
�� |
|
|
||
|
���������������ᣩ |
�� |
|
|
��3����6�֣�a.�ڽྻ���Թ��м���������������Һ������εμ�ϡ��ˮ�������������ɰ�ɫ��������Ӧ�����ӷ���ʽ�Ǣ�_______________________ �������μ�������ǡ���ܽ⣬����������Һ����Ӧ�����ӷ���ʽΪ��______________________��
b. ��a�����õ�������Һ�е���������������Һ����Ͼ��Ⱥ���ˮԡ�м���3min��5min���������Թ��ڱ����γ���������˷�Ӧ�Ļ�ѧ����ʽΪ�ۣ������ǵĽṹ��ʽ��G-CHO��ʾ����
(1) �ۢܣ� �٢ܣ� �ڣ� �ڢܡ�
(2)��
|
�� |
C |
b |
|
�� |
D |
a |
|
�� |
B |
c |
|
�� |
A |
b |
��3���� Ag++NH3��H2O=AgOH��+NH4+ ��AgOH+2NH3=[Ag(NH3)2]++OH-
��GCHO+2Ag(NH3)2OH GCOONH4+2Ag��+3NH3+H2O
GCOONH4+2Ag��+3NH3+H2O
����������1�����л������ܺ�����������Һ��Ӧ����±������ˮ�ⷴӦ����ȥ��Ӧ�����ǻ����Ȼ������������Դ��� �ۢܡ�
�ں���ȩ�����ܷ���������Ӧ�����Դ��� �٢ܡ�
���Ҵ������ǻ����ܷ�����ȥ��Ӧ��������Ӧ����ѡ�ڡ�
�ܿ����л�������ԭ��������жϡ�һ��ͬһ��̼ԭ���ϵ���ԭ������ͬ�ģ�ͬһ��̼ԭ�������ӵļ��ϵ���ԭ������ͬ�ģ����жԳ��Խṹ��������ƽ�澵�������������Ĺ�ϵ�����ݴ˿��жϢڢ���ȷ��
��2�����Ӻ��з��ǻ�����������ˮ������Ҫ��ȥ���еı��ӿ�ѡ������������Һ��Ȼ���Һ���Ҵ���ˮ���ܣ�����Ӧ������ʯ�ң�����Ҫ��ȡ�����������Ȼ��ƽ���֬�����Ƶ��ܽ���������ɣ���������������ˮ�����뱥��̼������Һ�����������Һ���ɡ�
��3������������Һ�����Ƽ�������Ӧ����ʽ����д��

 ��ǰ����ϵ�д�
��ǰ����ϵ�д���1����7�֣����������л��������Żش����и��ʣ�
�� 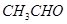 ��
�� 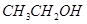 ��
��  ��
�� 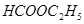
������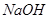 ��Һ��Ӧ����_________________��
��Һ��Ӧ����_________________��
������������Һ��Ӧ������������____________________��
�ۼ��ܷ�����ȥ��Ӧ�����ܷ���������Ӧ����___________________��
�ܺ˴Ź�������ͼ����3�����շ壬�����Ϊ3��2��1����___________��
��2������8�֣�Ϊ���Т����еij���ʵ�飨������Ϊ���ʣ�����Ӣ�����ѡ��һ���Լ����Ӣ�����ѡ��һ���ʵ�ʵ������������룬��������ں���Ĵ����ڡ�
| �� ����ʵ�� | �� �Լ� | �� �������� | �� | ||
| �� | �� | �� | |||
| �ٱ������ӣ� | A������Na2CO3��Һ B��NaCl���� C��NaOH��Һ D��CaO | a������ b����Һ c������������ d����ȡ | �� | | |
| ���Ҵ���ˮ�� | �� | | | ||
| �۷��������͡�ˮ�� | �� | | | ||
| ���������������ᣩ | �� | | | ||
b. ��a�����õ�������Һ�е���������������Һ����Ͼ��Ⱥ���ˮԡ�м���3min��5min���������Թ��ڱ����γ���������˷�Ӧ�Ļ�ѧ����ʽΪ�ۣ������ǵĽṹ��ʽ��G-CHO��ʾ����
��1�����������л��������Żش����и��ʣ�
��CH3CHO ��CH3CH2OH����CH3COOH����HCOOC2H5
������NaOH��Һ��Ӧ����______��
������������Һ��Ӧ������������______��
�ۼ��ܷ�����ȥ��Ӧ�����ܷ���������Ӧ����______��
�ܺ˴Ź�������ͼ����3�����շ壬�����Ϊ3��2��1����______��
��2��Ϊ���Т����еij���ʵ�飨������Ϊ���ʣ�����Ӣ�����ѡ��һ���Լ����Ӣ�����ѡ��һ���ʵ�ʵ������������룬��������ں���Ĵ����ڣ�
| �����ʵ�� | ���Լ� | ��������� | �� | ||
| �� | �� | �� | |||
| �ٱ������ӣ� | A������Na2CO3��Һ B��NaCl���� C��NaOH��Һ D��CaO | a������ b����Һ c������������ d����ȡ | �� | ||
| ���Ҵ���ˮ�� | �� | ||||
| �۷��������͡�ˮ�� | �� | ||||
| ���������������ᣩ | �� | ||||
b����a�����õ�������Һ�е���������������Һ����Ͼ��Ⱥ���ˮԡ�м���3min��5min���������Թ��ڱ����γ���������˷�Ӧ�Ļ�ѧ����ʽΪ�ۣ������ǵĽṹ��ʽ��G-CHO��ʾ����______��