��Ŀ����
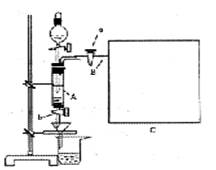
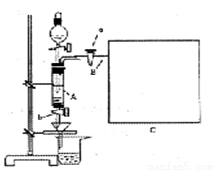
���Ȼ�������ѧ��ѧʵ�����бز����ٵ���Ҫ�Լ���ijͬѧ���÷���м��������ͭ�Ȳ������ᷴӦ�����ʣ����Ʊ�FeCl3��6H2O����ͬѧ��Ƶ�ʵ��װ����ͼ��ʾ��A�з���m�˷���м���ձ���ʢ�й�����ϡ���ᣬʵ��ʱ��a���ر�b���ӷ�Һ©������A�мӹ�����ϡ���ᣬ��ʱ��Һ��dz��ɫ���ٴ�b���й��ˣ����˽�����ȡ�ձ�����Һ������������ȣ�����������ˮ��ʹ����HNO3�ֽ⣬�ٽ��½ᾧ��FeCl3��6H2O���塣
��д���пհף�
��1����μ��װ��A�������ԣ� ��
��2���μ�����ʱ�����ַ�Ӧ���ʽ�֮ͬŨ�����������۷�ӦҪ�죬��ԭ���� ��
��3�����ձ�����Һ��������Ũ�����ٽ��½ᾧ���Ƶ�FeCl3��6H2O���壬������ֱ�������ᾧ�ķ������Ƶþ�������� ��
��4���ø÷��Ƶõľ�������������Fe��NO3��3��Ϊ���Ƶýϴ�����FeCl3��6H2O���ɽ��ձ��ڵ�ϡ���ỻ�� ��
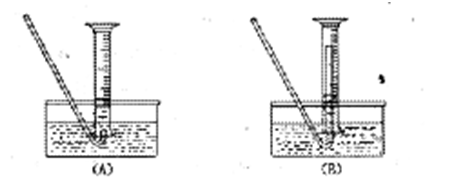
��5����Ҫ�����м�Ĵ��ȣ��ɲ����B���ų����������������������ʱ�����������ȴ������ʱ���У���������װ�òⶨ��B���ų��������������Ե�������Ͳ����ռ���������Ӧѡ�� ������ţ��������� ��
��1���رջ���a������b����Һ©��������ƿ�ǣ�����עˮ��©���¶˻��γ�һ��Һ����һ��ʱ�����Һ���������ƣ�˵�������Բ��ã���Һ��ֹͣ��������˵�������Ժá�����رշ�Һ©�������ͻ���b������a�����ܿ�B��һ�δ������ܵ��齺�ܾ�����ˮ���У���˫����סװ��A����ˮ���������ݲ������ſ�˫�ֲ��������γ�һ��Һ���������������á�����2�֣�
��2������������ͭ�γ�ԭ��ط�Ӧ���ӿ��˷�Ӧ���ʡ���2�֣�
��3����Ϊ�Ȼ�����ǿ��������ˮ�������������������ᣬ��������ʱʹHCl�ӷ����ˮ��ƽ�����ƣ���˵ò�����FeCl3��6H2O���壨2�֣�
��4��˫��ˮ��������ˮ������һ���������֣�����2�֣�
��5����B��2�֣�����ֹ������ȴʱ������������2�֣�
����:��