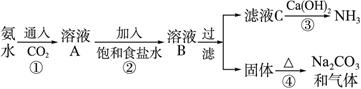
��Ŀ����
(15��)�Ȼ���ͭ��CuCl�����л��ϳɹ�ҵ��Ӧ�ýϹ㷺�Ĵ��������ǰ�ɫ��ĩ������ˮ���������Ҵ����ڿ����лᱻѸ�������������Ե�Ʒ�Һ����Ҫ��Cu2+��Fe3+�����Ʊ��Ȼ���ͭ�Ĺ�������ͼ���£�
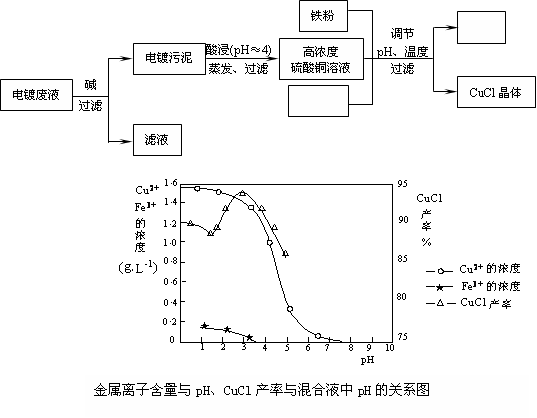
��ش��������⣺
��1������������Ҫ�ɷ��� ��д��ѧʽ����
��2�����ʱ������Ӧ�����ӷ���ʽ�� ��
��3������CuCl����ʱ�����pH�� ���ң�
��4�����ۡ��Ȼ��ơ�����ͭ����Һ�з�Ӧ����CuCl�����ӷ�Ӧ����ʽΪ��
��
��5��������CuCl����Ҫ��������ˮ�Ҵ�ϴ�ӣ�Ȼ����ո����ȴ���ܷ��װ����ո���ܷ��װ��Ŀ���� ��

��ش��������⣺
��1������������Ҫ�ɷ��� ��д��ѧʽ����
��2�����ʱ������Ӧ�����ӷ���ʽ�� ��
��3������CuCl����ʱ�����pH�� ���ң�
��4�����ۡ��Ȼ��ơ�����ͭ����Һ�з�Ӧ����CuCl�����ӷ�Ӧ����ʽΪ��
��
��5��������CuCl����Ҫ��������ˮ�Ҵ�ϴ�ӣ�Ȼ����ո����ȴ���ܷ��װ����ո���ܷ��װ��Ŀ���� ��
��(1��Cu(OH)2 Fe(OH)3����2�֣� ��2��Cu(OH)2+2H+= Cu2++2H2O��3�֣�
��3��3��2�֣� ��4��2Cu2++2Cl�D+ Fe��2CuCl��+ Fe2+��3�֣�
4.�ӿ��Ҵ���ˮ����������ֹCuCl������������3�֣�
��3��3��2�֣� ��4��2Cu2++2Cl�D+ Fe��2CuCl��+ Fe2+��3�֣�
4.�ӿ��Ҵ���ˮ����������ֹCuCl������������3�֣�
����1������������Ҫ�ɷ���Cu(OH)2 Fe(OH)3, ���ʱ������Ӧ�����ӷ���ʽ��Cu(OH)2+2H+= Cu2++2H2O, ����CuCl����ʱ�����pH��3���ҡ���ո���ܷ��װ��Ŀ���Ǽӿ��Ҵ���ˮ����������ֹCuCl������������

��ϰ��ϵ�д�
�����Ŀ
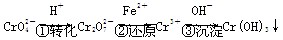
 Cr2O(��ɫ)��H2O
Cr2O(��ɫ)��H2O Na2S+2CO2��
Na2S+2CO2��