��Ŀ����
ʵ����������װ����ȡ����������������������ʵ�顣 �ش��������⣺
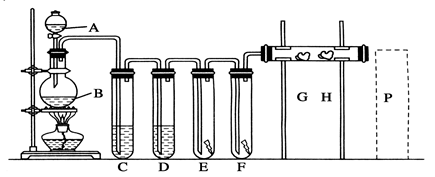
(1)A��B�����������ƣ�A_________________��B________________��
(2)ϴ��װ��C��Ϊ�˳�ȥCl2�е�HCl���壬Ӧ������Լ���________________��D��ŨH2SO4��������________________��
(3)E��Ϊ��ɫ�ɲ�����F��Ϊ��ɫʪ�������ɿ�����ɫ������ɫ���� _______������E��F��
(4��G�ǽ��е���KI��Һ������G�����������������______________��
H�ǽ���NaBr��Һ������H����Ӧ�����ӷ���ʽ��___________________________________________��
(5)P��Ϊβ������װ�ã� д��ʵ���������ռ���Һ��ȡCl2�����ӷ���ʽ�� ___________________��
(6)B�з�����Ӧ�Ļ�ѧ����ʽΪ_______________________ ����ʵ����ʹ��12 mol��L��1��Ũ����10 mL�������Ķ������̷�Ӧ�� ʵ�����ɵ�Cl2�����ʵ�������С��0��03 mol���Է������ܴ��ڵ�ԭ����______________________________________________________��

(1)A��B�����������ƣ�A_________________��B________________��
(2)ϴ��װ��C��Ϊ�˳�ȥCl2�е�HCl���壬Ӧ������Լ���________________��D��ŨH2SO4��������________________��
(3)E��Ϊ��ɫ�ɲ�����F��Ϊ��ɫʪ�������ɿ�����ɫ������ɫ���� _______������E��F��
(4��G�ǽ��е���KI��Һ������G�����������������______________��
H�ǽ���NaBr��Һ������H����Ӧ�����ӷ���ʽ��___________________________________________��
(5)P��Ϊβ������װ�ã� д��ʵ���������ռ���Һ��ȡCl2�����ӷ���ʽ�� ___________________��
(6)B�з�����Ӧ�Ļ�ѧ����ʽΪ_______________________ ����ʵ����ʹ��12 mol��L��1��Ũ����10 mL�������Ķ������̷�Ӧ�� ʵ�����ɵ�Cl2�����ʵ�������С��0��03 mol���Է������ܴ��ڵ�ԭ����______________________________________________________��
(1)��Һ©����Բ����ƿ (2)����ʳ��ˮ�� ��ȥ�����е�ˮ����
��3��F ��4����ɫ 2 Br����Cl2=2Cl����Br 2
��5��Cl2+2OH��=Cl��+ClO��+H2O
��6��MnO2��4HCl(Ũ�� MnCl2��Cl2����2H2O���ŷ�Ӧ�Ľ��У������Ȼ��⣬ͬʱ����ˮ�����Ȼ���ӷ���ʹ�����Ũ����С��ϡ������������̲���Ӧ��
MnCl2��Cl2����2H2O���ŷ�Ӧ�Ľ��У������Ȼ��⣬ͬʱ����ˮ�����Ȼ���ӷ���ʹ�����Ũ����С��ϡ������������̲���Ӧ��
��3��F ��4����ɫ 2 Br����Cl2=2Cl����Br 2
��5��Cl2+2OH��=Cl��+ClO��+H2O
��6��MnO2��4HCl(Ũ��
 MnCl2��Cl2����2H2O���ŷ�Ӧ�Ľ��У������Ȼ��⣬ͬʱ����ˮ�����Ȼ���ӷ���ʹ�����Ũ����С��ϡ������������̲���Ӧ��
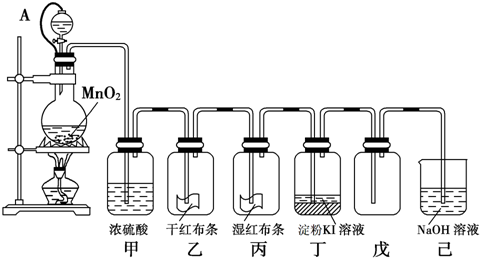
MnCl2��Cl2����2H2O���ŷ�Ӧ�Ľ��У������Ȼ��⣬ͬʱ����ˮ�����Ȼ���ӷ���ʹ�����Ũ����С��ϡ������������̲���Ӧ�������������1����װ��ͼʾ��֪��A�Ƿ�Һ©����B��Բ����ƿ��(2)��ŨHCl��MnO2�ڼ��������·�����ӦMnO2��4HCl(Ũ��
 MnCl2��Cl2����2H2O��ȡ����������Ũ�����лӷ��ԣ������������к�������HCl�����������ڱ���ʳ��ˮ�е��ܽ��С������Ϊ�˳�ȥCl2�е�HCl���壬Ӧ������Լ��DZ���ʳ��ˮ����ʱ��������������Ȼ����ˮ��������Ũ������������D��ŨH2SO4�������dz�ȥ�����е�ˮ������(3)����û��Ư���ԣ���������ˮʱ���߷�����Ӧ����HClO����Ư���ԡ����E�к�ɫ�ɲ�������ɫ��F�к�ɫʪ������ɫ��(4��G�ǽ��е���KI��Һ��������������KI������Ӧ��2 I����Cl2=2Cl����I 2��I 2�����۱�Ϊ��ɫ�����G�������������������ɫ��H�ǽ���NaBr��Һ���������ڻ��Cl2>Br 2������H����Ӧ�����ӷ���ʽ��2 Br����Cl2=2Cl����Br 2��(5)�����Ǵ�����Ⱦ��������ŷ�ǰӦ�ý���β������������������������Ӧ��������P��Ϊβ������װ�ã�ʵ���������ռ���Һ��ȡCl2�����ӷ���ʽ��Cl2+2OH��=Cl��+ClO��+H2O��(6)��B�з�����ʵ������ȡ�����ķ�Ӧ�Ļ�ѧ����ʽΪMnO2��4HCl(Ũ��
MnCl2��Cl2����2H2O��ȡ����������Ũ�����лӷ��ԣ������������к�������HCl�����������ڱ���ʳ��ˮ�е��ܽ��С������Ϊ�˳�ȥCl2�е�HCl���壬Ӧ������Լ��DZ���ʳ��ˮ����ʱ��������������Ȼ����ˮ��������Ũ������������D��ŨH2SO4�������dz�ȥ�����е�ˮ������(3)����û��Ư���ԣ���������ˮʱ���߷�����Ӧ����HClO����Ư���ԡ����E�к�ɫ�ɲ�������ɫ��F�к�ɫʪ������ɫ��(4��G�ǽ��е���KI��Һ��������������KI������Ӧ��2 I����Cl2=2Cl����I 2��I 2�����۱�Ϊ��ɫ�����G�������������������ɫ��H�ǽ���NaBr��Һ���������ڻ��Cl2>Br 2������H����Ӧ�����ӷ���ʽ��2 Br����Cl2=2Cl����Br 2��(5)�����Ǵ�����Ⱦ��������ŷ�ǰӦ�ý���β������������������������Ӧ��������P��Ϊβ������װ�ã�ʵ���������ռ���Һ��ȡCl2�����ӷ���ʽ��Cl2+2OH��=Cl��+ClO��+H2O��(6)��B�з�����ʵ������ȡ�����ķ�Ӧ�Ļ�ѧ����ʽΪMnO2��4HCl(Ũ�� MnCl2��Cl2����2H2O����ʵ����ʹ��12 mol/L��Ũ����10 mL�������Ķ������̷�Ӧ�� ʵ�����ɵ�Cl2�����ʵ�������С��0��03 mol������Ϊ���ŷ�Ӧ�Ľ��У������Ȼ��⣬ͬʱ����ˮ�����Ȼ���ӷ���ʹ�����Ũ����С��ϡ������������̲���Ӧ��
MnCl2��Cl2����2H2O����ʵ����ʹ��12 mol/L��Ũ����10 mL�������Ķ������̷�Ӧ�� ʵ�����ɵ�Cl2�����ʵ�������С��0��03 mol������Ϊ���ŷ�Ӧ�Ľ��У������Ȼ��⣬ͬʱ����ˮ�����Ȼ���ӷ���ʹ�����Ũ����С��ϡ������������̲���Ӧ��
��ϰ��ϵ�д�
 ���ѵ����Ԫ��ĩ���100��ϵ�д�
���ѵ����Ԫ��ĩ���100��ϵ�д� ��˼άС�ھ�100����ҵ��ϵ�д�
��˼άС�ھ�100����ҵ��ϵ�д� ��ʦָ��һ��ͨϵ�д�
��ʦָ��һ��ͨϵ�д�
�����Ŀ
 HCl+HClO��
HCl+HClO��