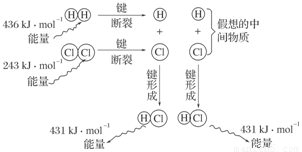
��Ŀ����
(8��) ��Ҫ��д�Ȼ�ѧ����ʽ��
(1) ��֪ϡ��Һ�У�1 mol H2SO4��NaOH��Һ
ǡ����ȫ��Ӧʱ���ų�114.6 kJ������
д����ʾH2SO4��NaOH��Ӧ���к��ȵ��Ȼ�ѧ����ʽ
_________________________________________________________________.
(2) 25�桢101 kPa�����³��ȼ��һ�����Ķ�������ų�����ΪQ kJ�����ⶨ�������ɵ�CO2ͨ����������ʯ��ˮ�в���25 g��ɫ������д����ʾ����ȼ���ȵ��Ȼ�ѧ����ʽ______________________________________________________��
(3) ��֪�����Ȼ�ѧ����ʽ��
��CH3COOH(l)��2O2(g)===2CO2(g)��2H2O(l) ��H1����870.3 kJ/mol
��C(s)��O2(g)===CO2(g)����H2����393.5 kJ/mol
��H2(g)��O2(g)===H2O(l)��H3����285.8 kJ/mol
д����C(s)��H2(g)��O2(g)��������CH3COOH(l)���Ȼ�ѧ����ʽ_________________________________________��
(1)H2SO4(aq)��NaOH(aq)===Na2SO4(aq)��H2O(l)����H����57.3 kJ/mol (2��)
(2)C4H10(g)��O2(g)===4CO2(g)��5H2O(l) ��H����16QkJ/mol (2��)
(3)2C(s)��2H2(g)��O2(g)===CH3COOH(l) ��H����488.3kJ/mol (4��)
����:��