��Ŀ����
��ǰ�ҹ����ٵ���ս�н�������������Դ����ʳ�ȷ�������⡣
(1)��ѧ�о��Ŀ���ܶ࣬��ٸ�Ч���ʵĺϳɣ�����ҩ����з������ڵͺ��������·ֽ�ˮ�Ƶ�����Ϊȼ�ϣ���Ѱ��������ɫ��Ⱦ����;��������������Ѫ�ܡ��ؽڵȣ������������������½��л����������߿������ܡ�ˮ�ܡ���ϫ�ܵȣ������Ƹ�Ч����ϴ�·ۡ�
�������ڽ����������__________(����ţ���ͬ)���������ڻ����������____________������������Դ�������____________������������ʳ�������____________��
(2)�г��ϳ��ֵġ���������ơ������ӵ�ʳ�Ρ������������ࡱ����Ʒ����Щ��Ʒ�еĸơ��⡢��Ӧ����Ϊ________��
A��ԭ�� B������ C������ D��Ԫ��
(3)ij����Ƭ�к�������ϸС�Ļ�ԭ���ۣ�ʳ��������Ƭ����������θҺ�б�������Σ��䷴Ӧ�Ļ�ѧ����ʽΪ___________________________________________________��
(1)�ڢݡ��ܢޢࡡ�ۢߡ��١�(2)D
(3)Fe��2HCl===FeCl2��H2��

��ϰ��ϵ�д�
 ���ɶ��ȫ���ƿؾ�ϵ�д�
���ɶ��ȫ���ƿؾ�ϵ�д�
�����Ŀ
 2C(g)(����Ӧ����)������ͼ������ȷ���ǣ� ��
2C(g)(����Ӧ����)������ͼ������ȷ���ǣ� ��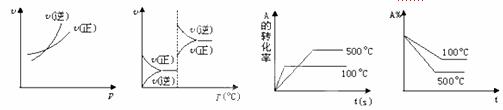
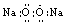
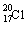 D.��ԭ�ӵĽṹʾ��ͼ��
D.��ԭ�ӵĽṹʾ��ͼ��