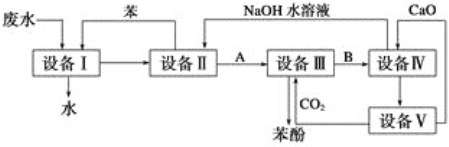
ћвƒњƒЏ»Ё
°Њћвƒњ°њƒ∆ЇЌЌ≠µƒµ•÷ Љ∞∆дїѓЇѕќп‘Џ…зїб µЉ ÷–”–„≈єгЈЇµƒ”¶”√°£
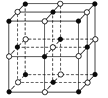
£®1£©NaClЊІћеµƒЊІ∞ыљбєє»зЌЉЋщ Њ£ђ√њЄцNaClµƒЊІ∞ы÷–Їђ”–µƒ“хјл„”µƒЄц эќ™__£ђ—фјл„”÷№ќІ„ољь«“µ»Њајлµ√“хјл„”µƒЄц эќ™__°£

£®2£©µвїѓƒ∆»№“ЇЇЌЅтЋбЌ≠»№“Їƒ№Јі”¶…ъ≥…“ї÷÷Ќ≠µƒµвїѓќпA£®∞„…Ђ≥Ѕµн£©£ђAµƒЊІ∞ы»з”“ЌЉЋщ Њ£ђ‘тAµƒїѓ—І љ «__£ђA÷–Ќ≠‘™ЋЎµƒїѓЇѕЉџќ™__°£
£®3£©ѕтЅтЋбЌ≠»№“Ї÷–µќЉ”∞±ЋЃїб…ъ≥…јґ…Ђ≥Ѕµн£ђ‘ўµќЉ”∞±ЋЃµљ≥ЅµнЄ’Ї√»Ђ≤њ»№љвњ…µ√µљ…ојґ…Ђ»№“Ї£ђЉћ–шѕт∆д÷–Љ”»лЉЂ–‘љѕ–°µƒ““іЉњ…“‘…ъ≥……ојґ…Ђµƒ[Cu(NH3)4]SO4°§H2O≥Ѕµн£ђЄ√ќп÷ ÷–µƒNH3Ќ®єэ___Љь”л÷––ƒјл„”Cu2£ЂљбЇѕ£ђNH3Ј÷„”÷–N‘≠„”µƒ‘”їѓЈљ љ «__°£”лNH3Ј÷„”ї•ќ™µ»µз„”ћеµƒ“ї÷÷—фјл„” «__°£
°Њір∞Є°њ4 6 CuI £Ђ1 ≈дќї sp3 H3O£ЂїтNa£ЂїтMg2£ЂїтAl3£Ђ
°Њљвќц°њ
£®1£©јы”√ЊщћѓЈ®Љ∆ЋгЊІ∞ы÷–Їђ”–µƒƒ∆јл„”ЇЌ¬»јл„” эƒњ£ї
£®2£©јы”√ЊщћѓЈ®Љ∆ЋгЊІ∞ы÷–Їђ”–µƒЌ≠јл„”ЇЌµвјл„” эƒњ£ђµ√µљAµƒїѓ—І љ£ђі”ґш»Јґ®Ќ≠µƒїѓЇѕЉџ£ї
£®3£©NH3÷–N‘≠„”ћбє©є¬ґ‘µз„”£ђCu2+ћбє©њ’ємµј£ђ–ќ≥…≈дќїЉь£їNH3Ј÷„”÷–є¬ґ‘µз„” эќ™1£ђ≥…Љь эќ™3£ђ”лNH3Ј÷„”ї•ќ™µ»µз„”ћеµƒ“ї÷÷ќҐЅ£Њя”–ѕаЌђµƒЉџµз„” эЇЌ‘≠„” э£ї
£®1£©ЇЏ…Ђ«т±н Њ¬»јл„”£ђ∞„…Ђ«т±н Њƒ∆јл„”£ђƒ∆јл„” эƒњ=1+12°Ѕ![]() =4£ђ¬»јл„” эƒњ=
=4£ђ¬»јл„” эƒњ=![]() =4£ђЋщ“‘√њЄцNaClЊІ∞ы÷–Їђ”–µƒNa+јл„”ЇЌClјл„”µƒ эƒњЈ÷±р «4°Ґ4£ђ“їЄцƒ∆јл„”÷№ќІ «6Єц¬»јл„”£ђЋщ“‘≈дќї эќ™6£ђє ір∞Єќ™£Ї4£ї6£ї
=4£ђЋщ“‘√њЄцNaClЊІ∞ы÷–Їђ”–µƒNa+јл„”ЇЌClјл„”µƒ эƒњЈ÷±р «4°Ґ4£ђ“їЄцƒ∆јл„”÷№ќІ «6Єц¬»јл„”£ђЋщ“‘≈дќї эќ™6£ђє ір∞Єќ™£Ї4£ї6£ї
£®2£©AµƒЊІ∞ы»зЌЉЋщ Њ,ЇЏ…Ђ«тµƒЄц э «![]() =4£ђ∞„«тќ™4Єц£ђЋщ“‘Aµƒїѓ—І љ «CuI£ђA÷–Ќ≠‘™ЋЎ «+1Љџ£ђє ір∞Єќ™£ЇCuI £ї£Ђ1£ї
=4£ђ∞„«тќ™4Єц£ђЋщ“‘Aµƒїѓ—І љ «CuI£ђA÷–Ќ≠‘™ЋЎ «+1Љџ£ђє ір∞Єќ™£ЇCuI £ї£Ђ1£ї
£®3£©NH3÷–N‘≠„”ћбє©є¬ґ‘µз„”£ђCu2+ћбє©њ’ємµј£ђґю’я–ќ≥…≈дќїЉь£ђNH3Ј÷„”÷–є¬ґ‘µз„” эќ™1£ђ≥…Љь эќ™3£ђ‘тN‘≠„”µƒ‘”їѓЈљ љќ™sp3£ї”лNH3Ј÷„”ї•ќ™µ»µз„”ћеµƒ“ї÷÷ќҐЅ£Њя”–ѕаЌђµƒЉџµз„” э8ЇЌ‘≠„” э4£ђќҐЅ£ќ™PH3їтH3O+µ»£ђє ір∞Єќ™£Ї≈дќї£їsp3£їPH3їтH3O+µ»°£

°Њћвƒњ°њ“їґ®ћхЉюѕ¬£ђґю—хїѓћЉњ…Їѕ≥…µЌћЉѕ©ћю£ђїЇљвќ¬ “–І”¶°Ґ≥дЈ÷јы”√ћЉ„ ‘і°£
£®1£©“—÷™£ЇҐўC2H4(g)+2O2(g)=2CO2(g)+2H2(g) ¶§H1
ҐЏ2H2(g)+O2(g)=2H2O(1) ¶§H2
ҐџH2O(1)=H2O(g) ¶§H3
Ґ№2CO2(g)+6H2(g)![]() C2H4(g)+4H2O(g) ¶§H4
C2H4(g)+4H2O(g) ¶§H4
‘т¶§H4=___(”√¶§H1°Ґ¶§H2°Ґ¶§H3±н Њ)°£
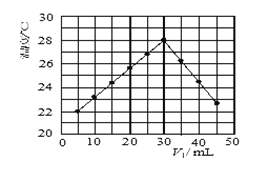
£®2£©Јі”¶Ґ№µƒЈі”¶ќ¬ґ»°ҐЌґЅѕ±»[![]() =x]ґ‘CO2∆љЇв„™їѓ¬ µƒ”∞ѕм»зЌЉЋщ Њ°£
=x]ґ‘CO2∆љЇв„™їѓ¬ µƒ”∞ѕм»зЌЉЋщ Њ°£

Ґўa__3£®ћо°∞>°±°Ґ°∞<°±їт°∞=°±£©£їM°ҐNЅљµгЈі”¶µƒ∆љЇв≥£ эKM__KN£®ћоћо°∞>°±°Ґ°∞<°±їт°∞=°±£©
ҐЏMµг““ѕ©ћеїэЈ÷ эќ™__£ї£®±£Ѕф2ќї”––І э„÷£©
Ґџ300°ж£ђЌщ6LЈі”¶»Ё∆ч÷–Љ”»л3molH2°Ґ1molCO2£ђЈі”¶10minіпµљ∆љЇв°£«у0µљ10min«в∆шµƒ∆љЊщЈі”¶Ћў¬ ќ™__£ї
£®3£©÷–њ∆‘ЇјЉ÷Ёїѓ—Іќпјн—–ЊњЋщ”√Fe3(CO)12/ZSM-5іяїѓCO2Љ”«вЇѕ≥…µЌћЉѕ©ћюЈі”¶£ђЋщµ√≤ъќпЇђCH4°ҐC3H6°ҐC4H8µ»Є±≤ъќп£ђЈі”¶єэ≥ћ»зЌЉ°£

іяїѓЉЅ÷–ћнЉ”Na°ҐK°ҐCu÷ъЉЅЇу£®÷ъЉЅ“≤∆ріяїѓ„ч”√£©њ…Єƒ±дЈі”¶µƒ—°‘с–‘£ђ‘Џ∆дЋыћхЉюѕаЌђ ±£ђћнЉ”≤їЌђ÷ъЉЅ£ђЊ≠єэѕаЌђ ±ЉдЇу≤вµ√CO2„™їѓ¬ ЇЌЄч≤ъќпµƒќп÷ µƒЅњЈ÷ э»зѕ¬±н°£
÷ъЉЅ | CO2„™їѓ¬ £®%£© | Єч≤ъќп‘ЏЋщ”–≤ъќп÷–µƒ’Љ±»£®%£© | ||
C2H4 | C3H6 | ∆дЋы | ||
Na | 42.5 | 35.9 | 39.6 | 24.5 |
K | 27.2 | 75.6 | 22.8 | 1.6 |
Cu | 9.8 | 80.7 | 12.5 | 6.8 |
Ґў”ыћбЄяµ•ќї ±ЉдƒЏ““ѕ©µƒ≤ъЅњ£ђ‘ЏFe3(CO)12/ZSM-5÷–ћнЉ”__÷ъЉЅ–Ієы„оЇ√£їЉ”»л÷ъЉЅƒ№ћбЄяµ•ќї ±ЉдƒЏ““ѕ©≤ъЅњµƒЄщ±Њ‘≠“т «__£ї
ҐЏѕ¬Ѕ–ЋµЈ®’э»Јµƒ «__£ї
a£ЃµЏҐ°≤љЋщЈі”¶ќ™£ЇCO2+H2![]() CO+H2O
CO+H2O
b£ЃµЏҐ°≤љЈі”¶µƒїоїѓƒ№µЌ”ЏµЏҐҐ≤љ
c£ЃіяїѓЉЅ÷ъЉЅ÷ч“™‘ЏµЌЊџЈі”¶°Ґ“мєєїѓЈі”¶їЈљЏ∆р„ч”√
d£ЃFe3(CO)12/ZSM-5 єCO2Љ”«вЇѕ≥…µЌћЉѕ©ћюµƒ¶§HЉх–°
e£ЃћнЉ”≤їЌђ÷ъЉЅЇу£ђЈі”¶µƒ∆љЇв≥£ эЄч≤їѕаЌђ
£®4£©2018ƒк£ђ«њЉо–‘µзіяїѓїє‘≠CO2÷∆““ѕ©—–Њњ»°µ√Ќї∆∆љш’є£ђ‘≠јн»зЌЉЋщ Њ°£

ҐўbЉЂљ”µƒ «ћЂ—фƒ№µз≥Ўµƒ__ЉЂ£ї
ҐЏ“—÷™PTFEљю≈ЁЅЋ±•ЇЌKCl»№“Ї£ђ«л–і≥ц“хЉЂµƒµзЉЂЈі”¶ љ__°£