ΧβΡΩΡΎ»ί
ΓΨΧβΡΩΓΩΆ≠ΒΞ÷ ΦΑΤδΜ·ΚœΈο‘ΎΚήΕύΝλ”ρ÷–ΕΦ”–÷Ί“ΣΒΡ”ΟΆΨΓΘ«κΜΊ¥π“‘œ¬Έ ΧβΘΚ
Θ®1Θ©≥§œΗΆ≠ΖέΩ…”ΟΉςΒΦΒγ≤ΡΝœΓΔ¥ΏΜ·ΦΝΒ»Θ§Τδ÷Τ±ΗΖΫΖ®»γœ¬ΘΚ
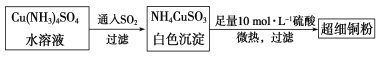
ΔΌNH4CuSO3÷–Ϋπ τ―τάκΉ”ΒΡΚΥΆβΒγΉ”≈≈≤Φ ΫΈΣ______ΓΘNΓΔOΓΔS»ΐ÷÷‘ΣΥΊΒΡΒΎ“ΜΒγάκΡή¥σ–ΓΥ≥–ρΈΣ______________(Χν‘ΣΥΊΖϊΚ≈)ΓΘ
ΔΎSO42Θ≠ΒΡΩ’ΦδΙΙ–ΆΈΣ________Θ§”κSO42Θ≠ΜΞΈΣΒ»ΒγΉ”ΧεΒΡ”–ΜζΖ÷Ή”ΒΡΜ·―ß ΫΈΣ_________ΓΘ
Θ®2Θ©Ρ≥―ß…ζ”ΟΝρΥαΆ≠»ή“Κ”κΑ±Υ°ΉωΝΥ“ΜΉι Β―ιΘ§CuSO4»ή“Κ÷–Φ”Α±Υ°…ζ≥…άΕ…Ϊ≥ΝΒμΘ§‘ΌΦ”Α±Υ°≥ΝΒμ»ήΫβΘ§ΒΟΒΫ…νάΕ…ΪΆΗΟς»ή“ΚΘ§ΉνΚσœρΗΟ»ή“Κ÷–Φ”»κ“ΜΕ®ΝΩ““¥ΦΘ§Έω≥ω[Cu(NH3)4]SO4ΓΛH2OΨßΧεΘ§«κΫβ ΆΦ”»κ““¥ΦΚσΈω≥ωΨßΧεΒΡ‘≠“ρ_______________ΘΜ‘ΎΗΟΨßΧε÷–¥φ‘ΎΒΡΜ·―ßΦϋΒΡ÷÷άύ”–_____________ΓΘ
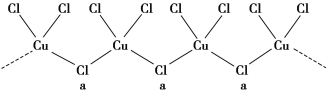
Θ®3Θ©¬»ΚΆΦΊ”κ≤ΜΆ§ΦέΧ§ΒΡΆ≠Ω……ζ≥…ΝΫ÷÷Μ·ΚœΈοΘ§Τδ“θάκΉ”ΨυΈΣΈόœό≥ΛΝ¥ΫαΙΙ(»γΆΦΥυ Ψ)Θ§aΈΜ÷Ο…œCl‘≠Ή”ΒΡ‘”Μ·ΙλΒάάύ–ΆΈΣ________ΓΘ“―÷ΣΤδ÷–“Μ÷÷Μ·ΚœΈοΒΡΜ·―ß ΫΈΣKCuCl3Θ§‘ρΝμ“Μ÷÷Μ·ΚœΈοΒΡΜ·―ß ΫΈΣ____________ΓΘ
Θ®4Θ©”ΟΨßΧεΒΡX…δœΏ―ή…δΖ®Ω…“‘≤βΒΟΑΔΖϋΦ”Β¬¬ό≥Θ ΐΒΡ÷ΒΓΘΕ‘Ϋπ τΆ≠ΒΡ≤βΕ®ΒΟΒΫ“‘œ¬ΫαΙϊΘΚΆ≠ΨßΑϊΈΣΟφ–ΡΝΔΖΫΉνΟήΕ―ΜΐΘ§±Ώ≥ΛΈΣ361 pmΓΘ”÷÷ΣΆ≠ΒΡΟήΕ»ΈΣ9.00 gΓΛcmΘ≠3Θ§‘ρΆ≠‘≠Ή”ΒΡ÷±ΨΕ‘ΦΈΣ__________pmΘ§«σΥψΑΔΖϋΦ”Β¬¬ό≥Θ ΐΒΡ±μ¥ο ΫΈΣ________ΓΘ
ΓΨ¥πΑΗΓΩΘ®1Θ©ΔΌ1s22s22p63s23p63d10(Μρ[Ar]3d10)ΘΜ N>O>S ΔΎ’ΐΥΡΟφΧε–Έ CCl4
Θ®2Θ©““¥ΦΖ÷Ή”±»Υ°Ζ÷Ή”ΦΪ–‘»θΘ§Φ”»κ““¥ΦΫΒΒΆ»ήΦΝΒΡΦΪ–‘Θ§¥”ΕχΦθ–Γ»ή÷ ΒΡ»ήΫβΕ»ΘΜ
άκΉ”ΦϋΓΔ≈δΈΜΦϋΓΔΙ≤ΦέΦϋ Θ®3Θ©sp3‘”Μ·ΘΜ K2CuCl3 Θ®4Θ©361/![]() pmΘΜ 4ΓΝ64/[9ΓΝ(3.61ΓΝ10Θ≠8)3]
pmΘΜ 4ΓΝ64/[9ΓΝ(3.61ΓΝ10Θ≠8)3]
ΓΨΫβΈωΓΩ
‘ΧβΖ÷ΈωΘΚΘ®1Θ©ΔΌNH4CuSO3÷–Ϋπ τ―τάκΉ”CuΘΪΘ§Ά≠ «29Κ≈‘ΣΥΊΘ§ΗυΨίΡήΝΩΉνΒΆ‘≠άμΤδΧ§‘≠Ή”ΒΡΒγΉ”≈≈≤Φ ΫΈΣ1s22s22p63s23p63d104s1Μρ[Ar]3d104s1Θ§ΗΟ‘≠Ή” ß»Ξ“ΜΗωΒγΉ” ±Θ§…ζ≥…―«Ά≠άκΉ”Θ§ ß»ΞΒΡΒγΉ” «ΉνΆβ≤ψΒγΉ”Θ§Υυ“‘―«Ά≠άκΉ”ΒΡΜυΧ§ΒΡΒγΉ”≈≈≤Φ ΫΈΣΘΚ1s22s22p63s23p63d10Μρ[Ar]3d10ΘΜ‘ΣΥΊ÷ήΤΎ¬…÷–Θ§Ά§“Μ÷ήΤΎ‘ΣΥΊΒΡΒΎ“ΜΒγάκΡήΥφΉ≈‘≠Ή”–ρ ΐΒΡ‘ω¥σΕχ‘ω¥σΘ§ΒΪΒΎΔρAΚΆΒΎΔσAΓΔΒΎΔθAΚΆΒΎΔωAΜΞΜΜΘ§Ά§“Μ÷ςΉε‘ΣΥΊΒΡΒΎ“ΜΒγάκΡήΥφΉ≈‘≠Ή”–ρ ΐΒΡ‘ω¥σΕχΦθ–ΓΘ§Υυ“‘ΤδΒΎ“ΜΒγάκΡή¥σ–ΓΈΣ NΘΨOΘΨSΓΘ
ΔΎSO42Θ≠άκΉ”÷–Φέ≤ψΒγΉ”Ε‘ ΐΈΣ4+![]() (6+2Θ≠4ΓΝ2)=4Θ§Ι¬ΒγΉ”Ε‘ ΐΈΣ0Θ§Υυ“‘ «’ΐΥΡΟφΧεΧε–ΆΘΜ‘≠Ή” ΐΚΆΦέΒγΉ” ΐΖ÷±πΕΦœύΒ»ΒΡ «Β»ΒγΉ”ΧεΘ§‘ρ”κΝρΥαΗυΜΞΈΣΒ»ΒγΉ”ΧεΒΡΖ÷Ή” «CCl4ΘΜ
(6+2Θ≠4ΓΝ2)=4Θ§Ι¬ΒγΉ”Ε‘ ΐΈΣ0Θ§Υυ“‘ «’ΐΥΡΟφΧεΧε–ΆΘΜ‘≠Ή” ΐΚΆΦέΒγΉ” ΐΖ÷±πΕΦœύΒ»ΒΡ «Β»ΒγΉ”ΧεΘ§‘ρ”κΝρΥαΗυΜΞΈΣΒ»ΒγΉ”ΧεΒΡΖ÷Ή” «CCl4ΘΜ
Θ®2Θ©““¥ΦΖ÷Ή”ΦΪ–‘±»Υ°Ζ÷Ή”ΦΪ–‘»θΘ§Φ”»κ““¥ΦΫΒΒΆ»ήΦΝΒΡΦΪ–‘Θ§¥”ΕχΦθ–Γ»ή÷ ΒΡ»ήΫβΕ»ΘΜ[Cu(NH3)4]SO4ΓΛH2O÷–ΝρΥαΗυάκΉ”ΚΆ[Cu(NH3)4]2+¥φ‘ΎάκΉ”ΦϋΘ§N‘≠Ή”ΚΆΆ≠‘≠Ή”÷°Φδ¥φ‘Ύ≈δΈΜΦϋΘ§NH3÷–HΚΆN÷°Φδ¥φ‘ΎΙ≤ΦέΦϋΘ§Υυ“‘[Cu(NH3)4]SO4ΓΛH2O÷–ΥυΚ§ΒΡΜ·―ßΦϋ”–Ι≤ΦέΦϋΓΔάκΉ”ΦϋΓΔ≈δΈΜΦϋΘΜ
Θ®3Θ©aΈΜ÷Ο…œCl‘≠Ή”≥…2ΗωΒΞΦϋΘ§Κ§”–2Ε‘Ι¬Ε‘ΒγΉ”Θ§‘”Μ·ΙλΒά ΐΈΣ4Θ§‘”Μ·ΙλΒάάύ–ΆΈΣsp3ΘΜ“Μ÷÷Μ·ΚœΈοΒΡΜ·―ß ΫΈΣKCuCl3Θ§Τδ÷–Ά≠‘ΣΥΊΈΣ+2ΦέΘ§Ι Νμ“Μ÷÷Μ·ΚœΈο÷–Ά≠ΈΣ+1ΦέΘ§CuCl3‘≠Ή”Ά≈ΒΡΜ·ΚœΦέΈΣ-2Θ§ΤδΜ·―ß ΫΈΣΘΚK2CuCl3ΘΜ
Θ®4Θ©Ά≠Οφ–ΡΝΔΖΫΉνΟήΕ―ΜΐΘ§±Ώ≥ΛΈΣ361 pmΘ§‘ρΆ≠‘≠Ή”ΒΡ÷±ΨΕ‘ΦΈΣ![]() pmΘΜΗυΨίΨßΑϊΒΡ±Ώ≥Λa=361pm=3.61ΓΝ10-8cmΘ§ΤδΧεΜΐΈΣΘ®3.61ΓΝ10-8cmΘ©3ΘΜΨßΑϊΒΡ÷ ΝΩ=9.00gΓΛcm3ΓΝΘ®3.61ΓΝ10-8cmΘ©3ΘΜΨßΑϊ÷–Κ§”–4ΗωCu‘≠Ή”Θ§Ι 64gΓΛmolΘ≠1=
pmΘΜΗυΨίΨßΑϊΒΡ±Ώ≥Λa=361pm=3.61ΓΝ10-8cmΘ§ΤδΧεΜΐΈΣΘ®3.61ΓΝ10-8cmΘ©3ΘΜΨßΑϊΒΡ÷ ΝΩ=9.00gΓΛcm3ΓΝΘ®3.61ΓΝ10-8cmΘ©3ΘΜΨßΑϊ÷–Κ§”–4ΗωCu‘≠Ή”Θ§Ι 64gΓΛmolΘ≠1=![]() ΓΝ9.00gΓΛcm3ΓΝΘ®3.61ΓΝ10-8cmΘ©3ΓΝNAΘ§ΫβΒΟNA=4ΓΝ64/[9ΓΝ(3.61ΓΝ10Θ≠8)3]molΘ≠1ΓΘ
ΓΝ9.00gΓΛcm3ΓΝΘ®3.61ΓΝ10-8cmΘ©3ΓΝNAΘ§ΫβΒΟNA=4ΓΝ64/[9ΓΝ(3.61ΓΝ10Θ≠8)3]molΘ≠1ΓΘ

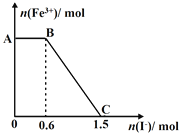
ΓΨΧβΡΩΓΩIΓΔ“―÷Σ‘ΎœΓ»ή“Κ÷–Θ§ΥαΗζΦνΖΔ…ζ÷–ΚΆΖ¥”Π…ζ≥…1molΥ° ±ΒΡΖ¥”Π»»Ϋ–Ήω÷–ΚΆ»»ΓΘœ÷άϊ”Οœ¬ΆΦΉΑ÷ΟΫχ––÷–ΚΆ»»ΒΡ≤βΕ®Θ§«κΜΊ¥πœ¬Ν–Έ ΧβΘΚ
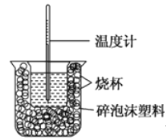
Θ®1Θ©ΗΟΆΦ÷–”–ΝΫ¥ΠΈ¥Μ≠≥ωΘ§ΥϋΟ«Ζ÷±π «______ΓΔ______ΓΘ
Θ®2Θ©Ήω“Μ¥ΈΆξ’ϊΒΡ÷–ΚΆ»»≤βΕ® Β―ιΘ§Έ¬Ε»ΦΤ–η Ι”Ο______¥ΈΓΘ
Θ®3Θ©Α―Έ¬Ε»ΈΣ13ΓφΘ§≈®Ε»ΈΣ1.0mol/LΒΡΥα»ή“ΚΚΆ1.1mol/LΒΡΦν»ή“ΚΗς50mLΜλΚœΘ®»ή“ΚΟήΕ»ΨυΈΣ1g/mLΘ©Θ§…ζ≥…»ή“ΚΒΡ±»»»»ίc=4.184J/(gΓφ)Θ§«α«αΫΝΕ·Θ§≤βΒΟΥαΦνΜλΚœ“ΚΒΡΈ¬Ε»±δΜ· ΐΨί»γœ¬ΘΚ
Ζ¥”ΠΈο | Τπ ΦΈ¬Ε»t1/Γφ | ÷’÷ΙΈ¬Ε»t2/Γφ | ÷–ΚΆ»» |
HCl+NaOH | 13 | 19.8 | ΓςH1 |
HCl+NH3H2O | 13 | 19.3 | ΓςH2 |
ΔΌ±»ΫœΓςH1ΚΆΓςH2ΒΡœύΕ‘¥σ–ΓΘΚΓςH1 ΓςH2Θ®ΧνΓΑΘΨΓ±ΓΑΘΦΓ±ΜρΓΑΘΫΓ±Θ©ΓΘ
ΔΎΝΫΉι Β―ιΫαΙϊ≤ν“λΒΡ‘≠“ρ « ΓΘ
Δέ–¥≥ωHClΚΆNH3H2OΖ¥”ΠΒΡ»»Μ·―ßΖΫ≥Χ ΫΘΚ ΓΘ
IIΓΔ»γœ¬ΆΦΥυ ΨΘΚ”ΟΝΫ’≈‘≤–Έ¬Υ÷ΫΖ÷±π’έΒΰ≥…¬©ΕΖΉ¥Θ§ΧΉ‘Ύ“ΜΤπΘ§ ΙΥΡ÷ήΕΦ”–ΥΡ≤ψΓΘΑ―ΡΎ≤ψ¬Υ÷Ϋ»Γ≥ωΘ§‘ΎΒΉ≤ΩΦτ“ΜΗω–ΓΩΉΘ§”ΟΥ°»σ ΣΘ§‘ΌΗζΝμ“Μ÷Ϋ¬©ΕΖΧΉ‘Ύ“ΜΤπΘ§Φή‘ΎΧζ»Π…œΘ§œ¬ΟφΖ≈÷Ο“Μ Δ…≥ΒΡ’τΖΔΟσΓΘΑ―5 gΗ…‘οΒΡ―θΜ·ΧζΖέΡ©ΚΆ2 g¬ΝΖέΜλΚœΨυ‘»Θ§Ζ≈‘Ύ÷Ϋ¬©ΕΖ÷–Θ§…œΟφΦ”…ΌΝΩ¬»ΥαΦΊ≤Δ‘ΎΜλΚœΈο÷–Φδ≤ε“ΜΗυΟΨΧθΘ§”Ο–ΓΡΨΧθΒψ»ΦΟΨΧθΓΘΙέ≤λΖΔ…ζΒΡœ÷œσΓΘΆ®Ιΐ“‘…œ Β―ι–≈œΔΘ§ΜΊ¥πœ¬Ν–Έ ΧβΘΚ
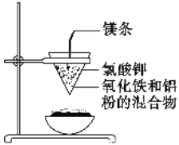
Θ®1Θ©–¥≥ωΗΟΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΘΚ__________Θ§ΗΟΖ¥”Π≥ΤΈΣ________Ζ¥”ΠΓΘ
Θ®2Θ©―Γ≥ωΗΟ Β―ιΒΡ Β―ιœ÷œσ(Χν–¥‘ΎΚαœΏ…œ)ΘΚ________ΓΘ
ΔΌΟΨΧθΨγΝ“»Φ…’ ΔΎΖ≈≥ω¥σΝΩΒΡ»»Θ§≤ΔΖΔ≥ω“Ϊ―έΒΡΙβΟΔΘ§Μπ–«ΥΡ…δ
Δέ÷Ϋ¬©ΕΖΒΡœ¬≤Ω±Μ…’¥© Δή”–Κλ»»Ή¥Χ§ΒΡ“Κ÷ι¬δ»κ’τΖΔΟσΡΎΒΡœΗ…≥…œΘ§“Κ÷ιά以Κσ±δΈΣΚΎ…ΪΙΧΧε
Θ®3Θ©–¥≥ωΗςΈο÷ ΒΡΉς”ΟΘΚ’τΖΔΟσ Δ…≥ΘΚ________ΘΜΟΨΧθΘΚ________ΘΜ¬»ΥαΦΊΘΚ________ΓΘ