ЬтФПФкШн
ЁОЬтФПЁПXЁЂYЁЂZОљЮЊЖЬжмЦкдЊЫизщГЩЕФЮяжЪЃЌЦфзЊЛЏЙиЯЕШчЯТЃК

ЧыЛиД№ЃК
ЃЈ1ЃЉШєXЮЊЕЛЦЩЋЙЬЬхЃЌZЪЧЕЅжЪЁЃ
ЂйзщГЩZЕФдЊЫидкжмЦкБэжаЕФЮЛжУЪЧ_____ЁЃ
ЂкШЁЩйСПXгыЫЎЗДгІЃЌЯТСаа№Ъіе§ШЗЕФЪЧ_____ЃЈЬюзжФИЃЉЁЃ
aЃЎИУЗДгІЪЧбѕЛЏЛЙдЗДгІ
bЃЎЯђЗДгІКѓЕФШмвКжаЕЮМгЗгЬЊЪдМСЃЌШмвКБфКь
cЃЎЕБга1 molXВЮМгЗДгІЪБЃЌЙВзЊвЦ2molЕчзг
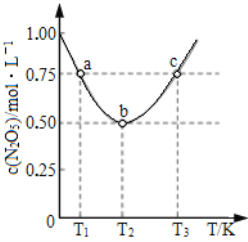
ЃЈ2ЃЉШєXгЩСНжждЊЫизщГЩЃЌЦфжавЛжждЊЫиЕФзюЭтВуЕчзгЪ§БШЕчзгВуЪ§Жр3ЃЌСэвЛжжЪЧЕиПЧжаКЌСПзюЖрЕФдЊЫиЁЃYгыXЕФзщГЩдЊЫиЯрЭЌЁЃ
ЂйXгыЫЎЗДгІЕФЛЏбЇЗНГЬЪНЮЊ_____ЁЃ
ЂкXгыO2ЛьКЯЭЈШыЫЎжаЃЌЧЁКУЩњГЩ4molZ,дђЭЈШыЕФXЮЊ____molЃЌO2ЮЊ____molЁЃ
ЁОД№АИЁПЕкЖўжмЦкЕкЂіAзх ab 3NO2+H2O=2HNO3+NO 4mol 1mol
ЁОНтЮіЁП
ИљОнЙ§бѕЛЏФЦгыЫЎЗДгІдРэЗжЮіНтД№ЃЛИљОнЖўбѕЛЏЕЊЕФЛЏбЇаджЪЗжЮіНтД№ЃЛИљОнЛЏбЇЗДгІЗНГЬЪНжаЕФЮяжЪЕФСПЕФМЦЫуЗжЮіНтД№ЁЃ
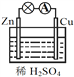
(1)ШєXЮЊЕЛЦЩЋЙЬЬхЃЌZЪЧЕЅжЪЃЌФмгыЫЎЗДгІЕФЕЛЦЩЋЙЬЬхЪЧNa2O2ЃЌгыЫЎЗДгІЩњГЩNaOHКЭO2ЃЌЫљвдXЪЧNa2O2ЃЌZЪЧO2ЃЌзщГЩZЕФдЊЫиМДOдЊЫидкжмЦкБэжаЕФЮЛжУЪЧЕкЖўжмЦкЕкЂіAзхЃЌИУЗДгІгаЛЏКЯМлБфЛЏЃЌЪЧбѕЛЏЛЙдЗДгІЃЌЗДгІЩњГЩNaOHЪЙШмвКЯдМюадЃЌГЪКьЩЋЃЌЕБга1 mol Na2O2ВЮМгЗДгІЪБЃЌЙВзЊвЦ1molЕчзгЃЌЙЪabе§ШЗЃЛ
ЙЪД№АИЮЊЃКЕкЖўжмЦкЕкЂіAзхЃЌabЃЛ
(2)ШєXгЩСНжждЊЫизщГЩЃЌЦфжавЛжждЊЫиЕФзюЭтВуЕчзгЪ§БШЕчзгВуЪ§Жр3ЃЌдђИУдЊЫиЮЊNдЊЫиЃЌСэвЛжжЪЧЕиПЧжаКЌСПзюЖрЕФдЊЫиЃЌдђИУдЊЫиЮЊOЁЃYгыXЕФзщГЩдЊЫиЯрЭЌЁЃИљОнЗДгІСїГЬПЩЭЦЖЯГіЃЌXЪЧNO2ЃЌYЪЧNOЃЌZЪЧHNO3ЃЌМДXгыЫЎЗДгІЕФЛЏбЇЗНГЬЪНЮЊЃК3NO2+H2O=2HNO3+NOЃЌЮяжЪXМДNO2ЃЌгыO2ЛьКЯЭЈШыЫЎжаЃЌЧЁКУЩњГЩ4molHNO3ЃЌЗДгІЗНГЬЪНЮЊЃК4NO2+O2+2H2O=4HNO3ЃЌМДЗДгІЩњГЩ4molHNO3ЪБЃЌашвЊЭЈШыNO2 4molЃЌашвЊЭЈШыбѕЦј1molЃЌ
ЙЪД№АИЮЊЃК4molЃЌ1molЁЃ

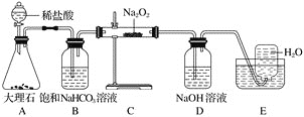
ЁОЬтФПЁПЯТЭМЪЧЪЕбщЪвжЦБИТШЦјВЂНјаавЛЯЕСаЯрЙиЪЕбщЕФзАжУ(МаГжЩшБИвбТд)ЁЃ
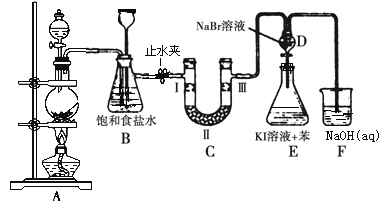
ЃЈ1ЃЉЪЕбщЪвжЦТШЦјЕФЛЏбЇЗНГЬЪНЮЊ____________________________ЁЃ
ЃЈ2ЃЉзАжУBжаБЅКЭЪГбЮЫЎЕФзїгУЪЧ______________ЃЛЧыаДГізАжУBЕФСэвЛИізїгУ_________________________________ЁЃ
ЃЈ3ЃЉзАжУCЕФЪЕбщФПЕФЪЧбщжЄТШЦјЪЧЗёОпгаЦЏАзадЃЌ ЮЊДЫCжаЂёЁЂЂђЁЂЂѓвРДЮЗХШыЮяжЪЕФзщКЯЪЧ________(ЬюБрКХ)ЁЃ
БрКХ | a | b | c | d |
Ђё | ИЩдяЕФгаЩЋВМЬѕ | ИЩдяЕФгаЩЋВМЬѕ | ЪЊШѓЕФгаЩЋВМЬѕ | ЪЊШѓЕФгаЩЋВМЬѕ |
Ђђ | МюЪЏЛв | ЙшНК | ХЈСђЫс | ЮоЫЎТШЛЏИЦ |
Ђѓ | ЪЊШѓЕФгаЩЋВМЬѕ | ЪЊШѓЕФгаЩЋВМЬѕ | ИЩдяЕФгаЩЋВМЬѕ | ИЩдяЕФгаЩЋВМЬѕ |
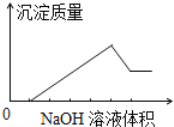
ЃЈ4ЃЉЩшМЦзАжУDЁЂEЕФФПЕФЪЧБШНЯТШЁЂфхЁЂЕтЕФЗЧН№ЪєадЁЃЗДгІвЛЖЮЪБМфКѓЃЌДђПЊЛюШћЃЌНЋзАжУDжаЩйСПШмвКМгШызАжУEжаЃЌеёЕДЃЌЙлВьЕНЕФЯжЯѓЪЧ____________________ЃЌИУЯжЯѓ_______(ЬюЁАФмЁБЛђЁАВЛФмЁБ)ЫЕУїфхЕФЗЧН№ЪєадЧПгкЕтЃЌдвђЪЧ_______________ЁЃ
ЃЈ5ЃЉгаШЫЬсГіЃЌзАжУFжаПЩИФгУзуСПЕФNa2SO3ШмвКЮќЪегрТШЃЌЪдаДГіЯргІЕФРызгЗДгІЗНГЬЪНЃК__________________ЁЃ
ЁОЬтФПЁПЯТСаЪЕбщжаЃЌЖдгІЕФЯжЯѓвдМАНсТлЖМе§ШЗЧвСНепОпгавђЙћЙиЯЕЕФЪЧ
бЁЯю | ЪЕбщ | ЯжЯѓ | НсТл |
A | НЋКьЩЋжНЬѕЗХШыЕНаТжЦТШЫЎжа | жНЬѕЭЪЩЋ | ТШЦјгаЦЏАзад |
B | НЋСђЫсЫсЛЏЕФH2O2ШмвКЕЮШыЕНFe(NO3)2ШмвКжа | ШмвКБфЛЦЩЋ | H2O2бѕЛЏадБШFe3+ЧП |
C | гУлсліЧЏМазЁвЛаЁПщгУЩАжНзаЯИДђФЅЙ§ЕФТСВдкОЦОЋЕЦЩЯМгШШ | ТСШлЛЏЧвЕЮТфЯТРД | ТСЕФШлЕуНЯЕЭ |
D | НЋNaIШмвКМгШыЕНAgClаќзЧвКжа | ВњЩњЛЦЩЋГСЕэ | Ksp(AgCl)>Ksp(AgI) |
A. AB. BC. CD. D