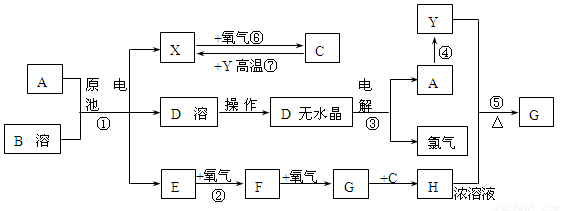
��Ŀ����
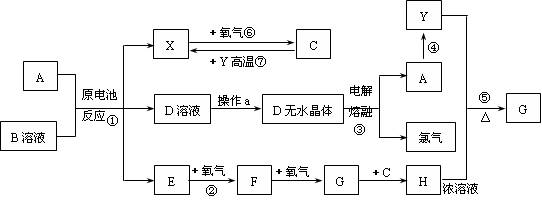
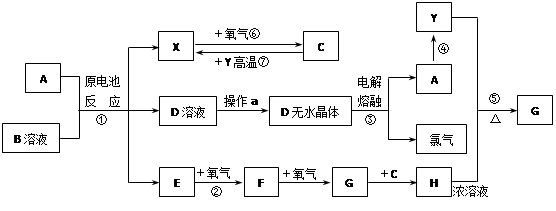
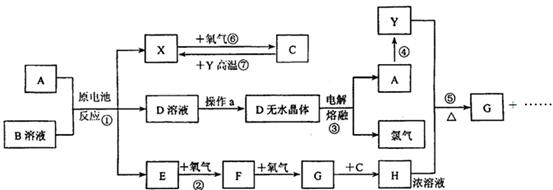
��֪AΪ����������X��YΪ�����ǽ�����X��E��F��G������Ϊ���壬CΪҺ�壬B��һ���Σ����ȼ��ֽ⣬�ڹ�ũҵ��������;�Ϲ㣨�类����ijЩ��صĵ���ʣ�������A��ʯī���缫��B��Ũ��Һ������ʣ�����ԭ��ء��й�����֮���ת����ϵ����ͼ����ע�⣺������Щ��Ӧ�����������������ﱻ��ȥ��
����д���пհף�
��1����Ӧ��ΪA��ij��������ȼ�գ����ɵ���Y��A��������䷴Ӧ����ʽΪ
______________________________________________________________________��
��2����D��Һ�Ʊ�D����ˮ����ġ�����a��Ϊ____________________________��
��3��E�ĵ���ʽΪ_________________________________��
��4����Ӧ�ݵĻ�ѧ����ʽΪ_______________________________________________��
��5��ԭ��ط�Ӧ���������ĵ缫��ӦʽΪ___________________________________.
��1��2Mg+CO2 2MgO+C�� ��2�֣�
��2����D��Һ��HCl���������ɣ���2�֣� ��3������2�֣�
��4����C+4HNO3 CO2��+4NO2��+2H2O ��2�֣�
��5��2NH4++2e- �� 2NH3��+H2�� ��2�֣�
����:��