题目内容
【题目】下图中A~J均为中学化学中常见的物质,它们之间有如下转化关系。其中A、D 为生活中常见金属单质。(反应过程中生成的水及其他产物已略去)
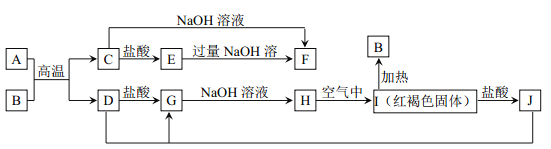
请回答以下问题:
(1)A 是_________,B是_________。(填化学式)
(2)A与B高温反应制得金属 D 的化学反应方程式为________________________。
(3)J的溶液可用来 “刻蚀”铜质电路板,在该刻蚀过程中,所发生反应的离子方程式为_________________________。
(4)检验J中阳离子的试剂是______________________。(写试剂名称)
【答案】(1)Al Fe2O3(2)2Al+Fe2O3![]() Al2O3+2Fe
Al2O3+2Fe
(3)2Fe3++Cu=2Fe2++Cu2+ (4)硫氰化钾
【解析】
试题分析:A、D 为生活中常见金属单质,A在高温下置换出D,且生成的C既能与盐酸反应,也能与氢氧化钠反应,所以反应是铝热反应,H在空气中变为红褐色,则H是氢氧化亚铁,因此A是铝,B是氧化铁,C是氧化铝,D是铁,E是氯化铝,F是偏铝酸钠,G是氯化亚铁,J是氯化铁。
(1)根据以上分析可知A 是Al,B是Fe2O3。
(2)A与B高温反应制得金属 D 的化学反应方程式为2Al+Fe2O3![]() Al2O3+2Fe。
Al2O3+2Fe。
(3)J的溶液可用来 “刻蚀”铜质电路板,在该刻蚀过程中,所发生反应的离子方程式为2Fe3++Cu=2Fe2++Cu2+。
(4)检验J中阳离子铁离子的试剂是硫氰化钾。

练习册系列答案
相关题目