��Ŀ����
��9�֣���֪A��B��C��D��E������������ͼ��ʾ��ת����ϵ�����ַ�Ӧ�P��Ӧ����δ�г���������ʱ��Ҫ�������������裩�������������о�����AԪ�ء�
��1����AΪ���嵥��
�� D�Ļ�ѧʽΪ ________________��
�� E��C�Ļ�ѧ����ʽΪ___________________________________________________
�� ��Cͨ��ij�ǽ������ʵ���Һ�У��ɷ���������ԭ��Ӧ��������ǿ�ᣬ�Ծ�һ��д����ѧ����ʽ_________________________________________________________��
��2����AΪ���嵥��
��B��C�Ļ�ѧ����ʽ________________________________________________________
�� E��C�����ӷ���ʽΪ__________________________________________________ ��
��1���� SO3 ��1�֣�
�� 2H2SO4(Ũ)+Cu ��CuSO4 ��SO2��+2H2O��2�֣�
�� Cl2��SO2+2H2O=2HCl+H2SO4��2�֣�
��2��
�� 4NH3+5 O2 �� 4NO+6H2O��2�֣�
����
�� 3Cu+8H+ +2NO3-=3Cu2++2NO��+4H2O��2�֣�
����:��

 �����Ծ���Ԫ���Ծ�ϵ�д�
�����Ծ���Ԫ���Ծ�ϵ�д�| A��Fe��Cu��Al��Ag | B��Al��Cu��Fe��Ag | C��Cu��Ag��Al��Fe | D��Ag��Al��Cu��Fe |
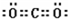
 ��֪A��B��C��D��E������������ͼ��ʾ��ת����ϵ�����ַ�Ӧ�P��Ӧ����δ�г���������ʱ��Ҫ�������������裩�������������о�����AԪ�أ�
��֪A��B��C��D��E������������ͼ��ʾ��ת����ϵ�����ַ�Ӧ�P��Ӧ����δ�г���������ʱ��Ҫ�������������裩�������������о�����AԪ�أ� ����ͨ������£���A��BΪ���ӣ�C��EΪ�����ӣ�DΪ�����ӣ����Ƕ�����10�����ӣ�B����A�����õ����ʿɵ����C��D��A��B��E��������Ӧ��ɵ�C��һ�ְ�ɫ��������ش�
����ͨ������£���A��BΪ���ӣ�C��EΪ�����ӣ�DΪ�����ӣ����Ƕ�����10�����ӣ�B����A�����õ����ʿɵ����C��D��A��B��E��������Ӧ��ɵ�C��һ�ְ�ɫ��������ش�