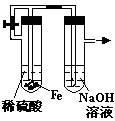
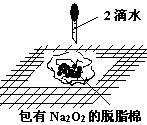
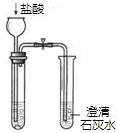
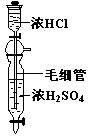
��Ŀ����
 ����������Ҫ�ɷ���
����������Ҫ�ɷ���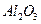 ����
����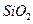 ��
��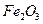 ��MgO�����ʣ�����ȡ���ֹ���Ʒ���������£�
��MgO�����ʣ�����ȡ���ֹ���Ʒ���������£�
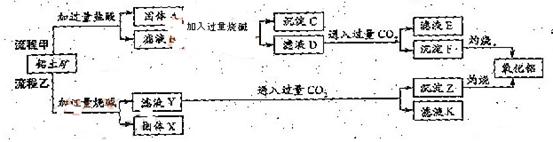
 ��ش��������⣺
��ش��������⣺ ��1�����̼������������Al3+�ķ���ʽΪ_________.
��1�����̼������������Al3+�ķ���ʽΪ_________. ��2�������Ҽ����ռ������Si
��2�������Ҽ����ռ������Si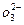 �����ӷ���ʽΪ________.
�����ӷ���ʽΪ________. ��3����֤��ҺB��
��3����֤��ҺB��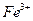 ����ȡ������Һ������________�����Լ����ƣ���
����ȡ������Һ������________�����Լ����ƣ��� ��4����ҺE��K�����ʵ���Ҫ�ɷ���________(�ѧʽ)��д������Һ��һ����;________
��4����ҺE��K�����ʵ���Ҫ�ɷ���________(�ѧʽ)��д������Һ��һ����;________ ��5����֪298Kʱ��
��5����֪298Kʱ��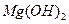 ���ݶȻ�����
���ݶȻ�����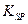 =5.6��
=5.6��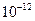 ,ȡ��������ҺB,����һ�������ռ�ﵽ������Һƽ�⣬���PH=13.00������¶��²�������Һ�е�
,ȡ��������ҺB,����һ�������ռ�ﵽ������Һƽ�⣬���PH=13.00������¶��²�������Һ�е�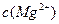 =_______.
=_______. ��1��Al2O3 + 6H��
��1��Al2O3 + 6H�� 2Al3�� +3H2O
2Al3�� +3H2O (2)SiO2 + 2OH��
(2)SiO2 + 2OH�� SiO32�� + H2O
SiO32�� + H2O (3)���軯�أ���������ء�������Һ�Ⱥ����𰸣�
(3)���軯�أ���������ء�������Һ�Ⱥ����𰸣� ��4��NaHCO3���ƴ���������ͷ۵Ⱥ�����
��4��NaHCO3���ƴ���������ͷ۵Ⱥ����� ��5��5.6��10-10mol/L
��5��5.6��10-10mol/L��1����HCl��Ӧ����Al3����ӦΪ��������Al2O3����2��SiO2��������NaOH������Na2SiO3����3������Fe3����������Ӧ�ܶ࣬����KSCN��Ѫ��ɫ���뱽������ɫ����OH���Ժ��ɫ�����ȡ���4������E��Kʱ��CO2���ǹ����ģ���Ӧ����NaHCO3����5�� ��
��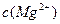 =5.6��10-12/(0.1)2=5.6��10-10��
=5.6��10-12/(0.1)2=5.6��10-10��
 ��
��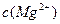 =5.6��10-12/(0.1)2=5.6��10-10��
=5.6��10-12/(0.1)2=5.6��10-10��
��ϰ��ϵ�д�
 ״Ԫ��ȫ��ͻ�Ƶ�����ϵ�д�
״Ԫ��ȫ��ͻ�Ƶ�����ϵ�д�
�����Ŀ