��Ŀ����
��ϩ��Ϊԭ�ϣ��ϳ�ijЩ�߾����·�����£�
��֪�� ����д��
����� ��
��
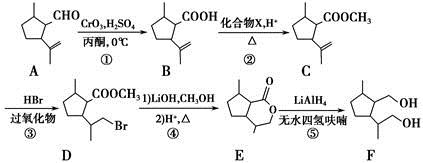
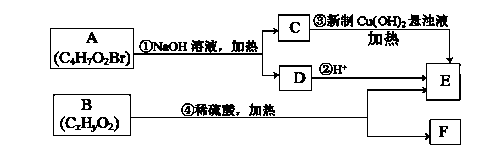
��1��CH3CH��CHCH3��������_______________��
��2��X�к��еĹ�������___________________��
��3��A��B�Ļ�ѧ����ʽ��__________________��
��4��D��E�ķ�Ӧ������_______________��
��5����Ϊ����F����NaHCO3��Ӧ����CO2��
�������й�˵����ȷ����_______________��
��Y��ͬ���칹���ж��֣�д�����ӽṹ�к�������������ͬ���칹��Ľṹ��ʽ____________��
��Z��W�Ļ�ѧ����ʽ��__________________��
��6���߾���H�Ľṹ��ʽ��_________________��

��֪��
 �����
����� ��
����1��CH3CH��CHCH3��������_______________��
��2��X�к��еĹ�������___________________��
��3��A��B�Ļ�ѧ����ʽ��__________________��
��4��D��E�ķ�Ӧ������_______________��
��5����Ϊ����F����NaHCO3��Ӧ����CO2��
�������й�˵����ȷ����_______________��
| A���л���Z�ܷ���������Ӧ | B���л���Y��HOCH2CH2OH��Ϊͬϵ�� |
| C���л���Y�ķе��B�� | D���л���F���뼺�������۳ɾۺ��� |
��Z��W�Ļ�ѧ����ʽ��__________________��
��6���߾���H�Ľṹ��ʽ��_________________��
��1��2-��ϩ
��2��̼̼˫�� ��ԭ��
��3��ClCH2CH2CH2CH2Cl +2NaOH
 CH2=CH��CH=CH2+ 2NaCl+2H2O��
CH2=CH��CH=CH2+ 2NaCl+2H2O����4����ȥ��Ӧ
��5����A D
�� HCOOCH2CH2CH3 HCOOCH��CH3��2 CH3COOCH2CH3 CH3CH2COOCH3
��
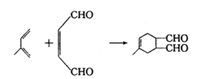
��6��

�����������1��CH3CH��CHCH3��������2-��ϩ����2��CH3CH��CHCH3��Cl2�ڹ��������·�������Cԭ���ϵ�ȡ����Ӧ����X��CH2ClCH��CHCH2Cl.�����X�к��еĹ�����Ϊ̼̼˫������ԭ�ӡ���3��X��H2��Ni�������������·����ӳɷ�Ӧ�õ�A��1,4--���ȶ���CH2ClCH2CH2CH2Cl ��A��NaOH���Ҵ���Һ���ȷ�����ȥ��ӦClCH2CH2CH2CH2Cl +2NaOH
 CH2=CH��CH=CH2+ 2NaCl+2H2O���õ�B��1,3--����ϩCH2=CH-CH=CH2��B����ϩ�����ӳɷ�Ӧ����
CH2=CH��CH=CH2+ 2NaCl+2H2O���õ�B��1,3--����ϩCH2=CH-CH=CH2��B����ϩ�����ӳɷ�Ӧ����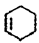 ��
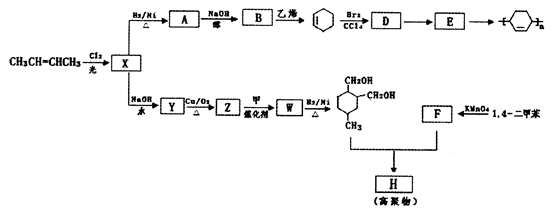
��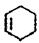 ��Br2��CCl4��Һ�����ӳɷ�Ӧ�õ�D��1,2-���廷����
��Br2��CCl4��Һ�����ӳɷ�Ӧ�õ�D��1,2-���廷���� ����4��D��NaOH���Ҵ���Һ������ȥ��Ӧ�õ�E��������ϩ
����4��D��NaOH���Ҵ���Һ������ȥ��Ӧ�õ�E��������ϩ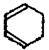 ��E�����Ӿ۷�Ӧ�õ�
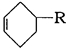
��E�����Ӿ۷�Ӧ�õ� ����5��X��CH2ClCH��CHCH2Cl����NaOH��ˮ��Һ����ȡ����Ӧ����Y��CH2��OH��CH��CHCH2OH;Y��O2��Cu������������´��ǻ�����������Ӧ�õ�Z��OHCCH��CHCHO��Z���Ӧ�IJ���W��������Ni�������������·����ӳɷ�Ӧ����Ϊ
����5��X��CH2ClCH��CHCH2Cl����NaOH��ˮ��Һ����ȡ����Ӧ����Y��CH2��OH��CH��CHCH2OH;Y��O2��Cu������������´��ǻ�����������Ӧ�õ�Z��OHCCH��CHCHO��Z���Ӧ�IJ���W��������Ni�������������·����ӳɷ�Ӧ����Ϊ ��������Ϊ2-��-1,3-����ϩ
��������Ϊ2-��-1,3-����ϩ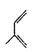 ��WΪ��
��WΪ��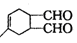 ��1,4--���ױ������Ը��������Һ����ΪF���Ա������ᣬF����
��1,4--���ױ������Ը��������Һ����ΪF���Ա������ᣬF����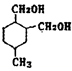 ����������Ӧ�õ��߾���H��
����������Ӧ�õ��߾���H��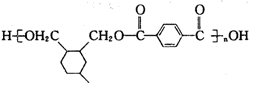 ����A. �л���Z OHCCH��CHCHO�к���ȩ������˿��Է���������Ӧ����ȷ��B. �л���Y CH2��OH��CH��CHCH2OH��HOCH2CH2OH���ܶ�����2�����ǻ�����ǰ����������˫��������û�У���˲���ͬϵ�����C���л���Y CH2��OH��CH��CHCH2OH�ķе��B��1,3--����ϩCH2=CH-CH=CH2����Է�����������Է�������Խ�����ʵ��۷е��Խ�ߡ����Y�ķе��B�ĸߡ�����D���л���F�Ա������Ậ�������Ȼ�������������������������˶����ܷ������۷�Ӧ�����ۺ����ȷ����CH2��OH��CH��CHCH2OH�ж���ͬ���칹�壬���з��ӽṹ�к�������������ͬ���칹����HCOOCH2CH2CH3�� HCOOCH��CH3��2 ��CH3COOCH2CH3��CH3CH2COOCH3����Z��W�Ļ�ѧ����ʽ��
����A. �л���Z OHCCH��CHCHO�к���ȩ������˿��Է���������Ӧ����ȷ��B. �л���Y CH2��OH��CH��CHCH2OH��HOCH2CH2OH���ܶ�����2�����ǻ�����ǰ����������˫��������û�У���˲���ͬϵ�����C���л���Y CH2��OH��CH��CHCH2OH�ķе��B��1,3--����ϩCH2=CH-CH=CH2����Է�����������Է�������Խ�����ʵ��۷е��Խ�ߡ����Y�ķе��B�ĸߡ�����D���л���F�Ա������Ậ�������Ȼ�������������������������˶����ܷ������۷�Ӧ�����ۺ����ȷ����CH2��OH��CH��CHCH2OH�ж���ͬ���칹�壬���з��ӽṹ�к�������������ͬ���칹����HCOOCH2CH2CH3�� HCOOCH��CH3��2 ��CH3COOCH2CH3��CH3CH2COOCH3����Z��W�Ļ�ѧ����ʽ��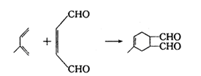 ����6���߾���H�Ľṹ��ʽΪ
����6���߾���H�Ľṹ��ʽΪ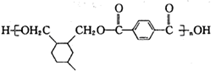

��ϰ��ϵ�д�
�����Ŀ
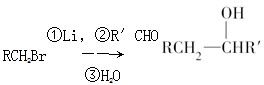
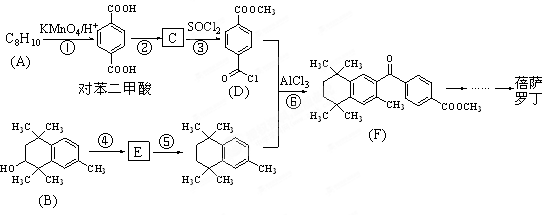
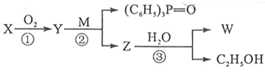
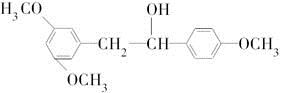 �Ǻϳɿ���ҩ�����«�����м��壬��д����
�Ǻϳɿ���ҩ�����«�����м��壬��д����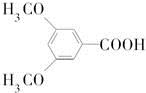 ��
��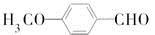 ��CH3OHΪԭ���Ʊ��û�����ĺϳ�·������ͼ(���Լ�����)���ϳ�·������ͼʾ�����£�
��CH3OHΪԭ���Ʊ��û�����ĺϳ�·������ͼ(���Լ�����)���ϳ�·������ͼʾ�����£�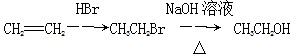
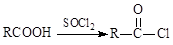
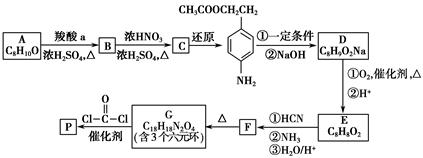
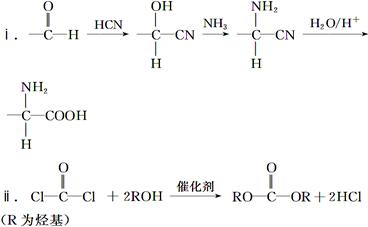
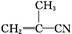
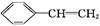

 CH��CN
CH��CN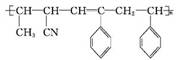 �ĸ߷��Ӳ��ϵ���ȷ���Ϊ�� ��
�ĸ߷��Ӳ��ϵ���ȷ���Ϊ�� ��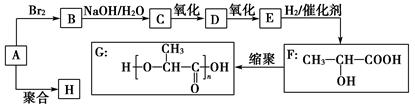

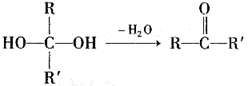
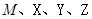 �ֱ����һ���л���ϳɹ�������������ͷ�Ӧ��������ȥ��
�ֱ����һ���л���ϳɹ�������������ͷ�Ӧ��������ȥ��