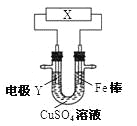
��Ŀ����
����Ŀ��ij��Һ�п��ܴ������ڣ�H+��Na+��Fe2+��Al3+��NH4+��SO42-��Cl-�еļ������ӣ��ֽ���������ʵ�飺
�� ȡ����Һ10.0mL���������Ba(NO3)2��Һ��������ɫ����x����ɫ����a��a������������Ϊ����ɫ����b�����ˡ�
�� ������õ���Һ�м������NaOH��Һ���������ɫ����y�����ˣ�������Һ����ɫ�̼�������c���ɣ��ò�˿պȡ��Һ���ڻ��������գ�����ʻ�ɫ��
�� ������õ���Һ��ͨ�����CO2�����а�ɫ����z���ɡ�
�����ƶϴ������
A. ԭ��Һһ��ֻ���У�Na+��Fe2+��Al3+��SO42-
B. ʵ��� �ĺ���ɫ����b�д��ڣ�2NO2![]() N2O4��
N2O4��
C. ʵ��� �в�������y�ķ�Ӧ��Fe3++3OH-=Fe(OH)3��
D. ʵ��� ��CO2�Ⱥ�ֱ���NaOH(aq)��NaAlO2(aq)��Ӧ
���𰸡�A
���������١�ȡ����Һ10.0mL���������Ba(NO3)2��Һ��������ɫ����x����ɫ����a��a������������Ϊ����ɫ����b�����ɫ����ΪBaSO4��aΪNO��bΪNO2������Һ��һ����H+��Fe2+��SO42-���ڡ�������õ���Һ�м������NaOH��Һ���������ɫ����y�����ˣ�������Һ����ɫ�̼�������c������y������������c�ǰ�������һ����NH4+���ò�˿պȡ��Һ���ڻ��������գ�����ʻ�ɫ����NaOH��Һ������ȷ��ԭ��Һ�Ƿ�Na+������������õ���Һ��ͨ�����CO2�����а�ɫ����z���ɣ���ɫ����Ϊ������������һ����Al3+��A��������������֪��һ����H+��Fe2+��Al3+��NH4+��SO42-������ȷ���Ƿ�Na+��Cl-��A������B��ʵ��� �ĺ���ɫ����b�д��ڣ�2NO2![]() N2O4��B��ȷ��C��ʵ��� �в�������y�ķ�Ӧ��Fe3++3OH-=Fe(OH)3����C��ȷ��D������NaOH�������Һ�ƫ��������Һ�����к͵��Ⱥ�˳���֪��ʵ�����CO2�Ⱥ�ֱ���NaOH(aq)��NaAlO2(aq)��Ӧ��D��ȷ����ѡA��
N2O4��B��ȷ��C��ʵ��� �в�������y�ķ�Ӧ��Fe3++3OH-=Fe(OH)3����C��ȷ��D������NaOH�������Һ�ƫ��������Һ�����к͵��Ⱥ�˳���֪��ʵ�����CO2�Ⱥ�ֱ���NaOH(aq)��NaAlO2(aq)��Ӧ��D��ȷ����ѡA��

����Ŀ����ѧ����ᡢ����������أ������й����ʵ���;��ԭ��������ȷ����
ѡ�� | ���ʵ���; | ԭ���ͷ��� |
A | Ư�ۿ�����ʳƷƯ�� | Ư����ʹʳƷ�е�ɫ����ɫ |
B | Al(OH)3���к�θ�� | Al(OH)3����θҺ�е��ᷢ���кͷ�Ӧ |
C | Na2O2���������� | Na2O2�ڳ������ֽܷ�ų�O2 |
D | ����������ˮ�� | ��������ˮ��������ɱ�� |
A. A B. B C. C D. D