��Ŀ����
I. (6��)��������Ʒ���豸�����մɲ;� ��ש�� ��ˮ��·�� ���Ŵ����� ��ˮ����Ƭ ��ʯӢ�ӱ� ��������� ���̫���ܵ�� ����ά ������оƬ
(1)�õ��赥�ʵ���_____________
(2)���ò���ΪSiO2��Ҫ�õ�SiO2����__________________
(3)���ò���Ϊ�����ε���________________ _��
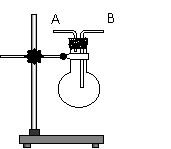
II��(6��)��ͼ���߿��е�װ�ÿ���������Ũ������ľ̿���ڼ��������·�Ӧ��������������������д���пհף�

װ���Тټ����������_______ ���ڵ������� ��
�ۿ���֤���������� ��Ũ������ľ̿���ڼ��������·�Ӧ�Ļ�ѧ����ʽ�ǣ�_____________ ��
 III����4�֣�������ͼ��ʾ��װ���ռ�����6�����壺
III����4�֣�������ͼ��ʾ��װ���ռ�����6�����壺
��CO2����C12����NO2����SO2 ����NH3����NO
(1)����ƿ�Ǹ���ģ���B�ڽ��������ռ��������� ��
(2)������ƿ�ڳ���ˮ���ռ�NO����,������Ӧ�� �ڽ��롣
I. (1)��⣨2�֣� (2)�ݢޢߢ� ��2�֣� (3)�٢ڢۢܣ�2�֣�
II��H2O ��1�֣�������ͳ�ȥSO2����֤��SO2�ѳ�ȥ��2�֣� �� CO2 ��1�֣��� 2H2SO4��Ũ��+C==∆== 2SO2��+CO2��+2H2O ��2�֣�
III.��1���٢ڢۢܣ�3�֣�����2�� A ��1�֣�

 �����������һ��һ��ϵ�д�
�����������һ��һ��ϵ�д�
 �����Ҫ�ϳ�
�����Ҫ�ϳ� ���õ�ԭʼԭ�Ͽ�����
���õ�ԭʼԭ�Ͽ�����
 F���������������У���ṹ��ʽΪ_______________��
F���������������У���ṹ��ʽΪ_______________��

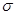 ����̼��֮��Ĺ��ۼ�������______________������֮���γɵĻ�ѧ����_______________��
����̼��֮��Ĺ��ۼ�������______________������֮���γɵĻ�ѧ����_______________��