��Ŀ����
����Ŀ���ߴ�MnCO3���Ʊ������ܴ��Բ��ϵ���Ҫԭ�ϡ�ʵ������MnO2Ϊԭ���Ʊ������ߴ�MnCO3�IJ�������������ʾ��
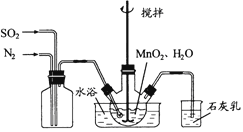
��.�Ʊ�MnSO4��Һ��
��������ƿ�м���4.35gMnSO4��������ˮ�����裬ͨ��SO2��N2������壬��Ӧ3h��ֹͣͨ��SO2��������ӦƬ�̣����ˡ�ϴ�ӣ���MnO2��Һ����N2�����뷴Ӧ��
��1����ȡMnSO4�Ļ�ѧ����ʽΪ_______________��
��2����Ӧ�����У�ΪʹSO2������ת����ȫ���ڲ��ı��ҺͶ�ϵ������£��ɲ�ȡ�ĺ�����ʩ��________________��
��3��ˮԡ���ȵ��ŵ���____________��
��.�Ʊ��ߴ�MnCO3���壺
��֪MnCO3������ˮ���Ҵ�����ʪʱ�ױ�����������100�濪ʼ�ֽ⣻pH=7.7ʱMn(OH)2��ʼ������
ʵ�鲽��: �ٽ�����MnSO4��Һ��20.0mL 2.0mol/L��Na2CO3��Һ��ϣ���ַ�Ӧ;�ڹ��ˣ�������ˮϴ��2~3�� ;��������C2H5OHϴ��;�ܵ��� (����100��) ����ù���3.45g�� (1) MnSO4��Һ��Na2CO3��Һ��ϵ���ȷ����Ϊ__________��(����ĸ����)
a. ��Na2CO3��Һ�����μӵ�MnSO4��Һ�У��ӱ߽���
b. ��MnSO4��Һ�����μӵ�Na2CO3��Һ�У��ӱ߽���
c. ��Na2CO3��ҺѸ�ٵ��뵽MnSO4��Һ�У�����ֽ���
d. ��MnSO4��ҺѸ�ٵ��뵽Na2CO3��Һ�У�����ֽ���
(2)����MnCO3�����Ƿ�ϴ�Ӹɾ��ķ���Ϊ______________��
(3) ������C2H5OHϴ�ӵ�Ŀ����_______________��
(4) MnCO3�IJ���Ϊ_____________��
���𰸡� MnO2+SO2=MnSO4����MnO2+H2SO4=MnSO4+H2O�� �����ʵ���ˮԡʪ�ȣ�����ͨ��������ٶȣ���СSO2�ڻ��������N2���еı��� ���Ⱦ��ȣ�������ʪ�� a ȡ���һ��ϴ��Һ�������Թ��У��μ�BaCl2���ף����������������˵����ϴ�Ӹɾ�������˵��δϴ�Ӹɾ� �Ҵ��е����100�棬���ڵ��¸�����MnCO3���ֽ� 75%
��������������Ҫ�������ʵ������MnO2Ϊԭ���Ʊ������ߴ�MnCO3ʵ������ۡ�
��.��1����ȡMnSO4�Ļ�ѧ����ʽΪMnO2+SO2=MnSO4��
��2����Ӧ�����У�ΪʹSO2������ת����ȫ���ڲ��ı��ҺͶ�ϵ������£��ɲ�ȡ�ĺ�����ʩ�п����ʵ���ˮԡ�¶ȣ�����ͨ��������ٶȣ���СSO2�ڻ��������N2���еı�����
��3��ˮԡ���ȵ��ŵ������Ⱦ��ȣ��������¶ȡ�
��.(1) ��ΪpH=7.7ʱMn(OH)2��ʼ��������Na2CO3��Һ��pH>7.7������Ӧ��Na2CO3��Һ�����μӵ�MnSO4��Һ�У��ӱ߽��裬��ѡa��
(2)����MnCO3�����Ƿ�ϴ�Ӹɾ����Ǽ����Ƿ����Na+��![]() ������
������![]() ������飬����Ϊ��ȡ���һ��ϴ��Һ�������Թ��У��μ�BaCl2��Һ�����������������˵����ϴ�Ӹɾ�������˵��δϴ�Ӹɾ���
������飬����Ϊ��ȡ���һ��ϴ��Һ�������Թ��У��μ�BaCl2��Һ�����������������˵����ϴ�Ӹɾ�������˵��δϴ�Ӹɾ���
(3) ������C2H5OHϴ�ӵ�Ŀ�����Ҵ��е����100�������ڵ��¸�����MnCO3���ֽ⡣
(4) 4.35g��0,05molMnO2��������ˮ��SO2��Ӧ�ú���0.05molMnSO4����Һ��20.0mL 2.0mol/L��Na2CO3��Һ����0.04molNa2CO3��������MnSO4��Һ��20.0mL 2.0mol/L��Na2CO3��Һ��ϣ���ַ�Ӧ�������ϵõ�0.04molMnCO3������MnCO3�IJ���Ϊ3.45g/[(0.04��115)g]=75%��

 ��ѧ����ͬ����ϰϵ�д�
��ѧ����ͬ����ϰϵ�д� ��ǰ�κ�ͬ����ϰϵ�д�
��ǰ�κ�ͬ����ϰϵ�д� ����С��ҵϵ�д�
����С��ҵϵ�д� �Ƹ�С״Ԫ����������ϰ��ϵ�д�
�Ƹ�С״Ԫ����������ϰ��ϵ�д�����Ŀ���������⡿�����µ����[Ca(IO3)2]��һ�ְ�ɫ���壬����ˮ���������Ҵ������������ᣬ��Ŀǰ�㷺ʹ�õļ��ܲ������ܲ��Ƶ�����ʳƷ�ʹ������Ӽ������Ʊ�ԭ�����£�
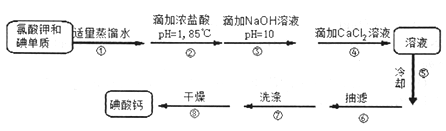
��.����Ƶ��Ʊ�
(1)ijͬѧ��Ƶ�ʵ��װ������ͼ��ʾ(�г�װ����ʡ��)������ڵļ��ȷ�ʽΪ_________��
(2)�������ȴ�ȵı�����Һû���������壬Ϊ�˵õ�������Բ�ȡ��һ�ִ�ʩ��________��
(3)�����ϴ�ӵľ��������____________��
��.��Ʒ���Ȳⶨ
ȷ��ȡ0.5000 g��Ʒ���ữ�ܽ⣬������250 mL,����ȡ��25.00 mL��������ƿ�У�����������KI��ַ�Ӧ����0.04000 mol��L-1�����������Һ�ζ����յ㣬�ظ����ϲ��裬�����ʵ���������£�
1 | 2 | 3 | |
�ζ���ʼ����/mL | 1.52 | 1.16 | 0.84 |
�ζ���ֹ����/mL | 31.50 | 31.18 | 30.84 |
��֪��2Na2S2O3 + I2= Na2S4O6 + 2NaI
(4)����KIʱ������Ӧ�����ӷ���ʽ��_____________��
(5)�ζ�ʱ���õ�ָʾ����_________________.
(6)�������Ʒ�Ĵ���Ϊ________________��