��Ŀ����
ʵ���ҳ����ü�ȩ���ⶨ��NH4��2SO4��Ʒ�е��������������䷴Ӧԭ��Ϊ��4NH4++6HCHO�T3H++6H2O+��CH2��6N4H+[�ζ�ʱ��1mol��CH2��6N4H+��1mol H+�൱]��Ȼ����NaOH����Һ�ζ���Ӧ���ɵ��ᣮij��ȤС���ü�ȩ������������ʵ�飺
������ȡ��Ʒ1.5g��
�������Ʒ�ܽ���250mL����ƿ�У����ݣ����ҡ�ȣ�
�������ȡ25.00mL��Ʒ��Һ��250mL��ƿ�У�����10mL 20%�����Լ�ȩ��Һ��ҡ�ȡ�����5min����1��2�η�̪��Һ����NaOH����Һ�ζ����յ㣮�����������������ظ�2�Σ�
��1����������������Ƿ���ȷ
��2�����ݲ������գ�
�ټ�ʽ�ζ���������ˮϴ�Ӻ�ֱ�Ӽ���NaOH����Һ���еζ���������Ʒ�е�����������
����ƿ������ˮϴ�Ӻ�ˮδ��������ζ�ʱ��ȥNaOH����Һ�����
�۵ζ�ʱ�ߵα�ҡ����ƿ���۾�Ӧ�۲�
A���ζ�����Һ��ı仯
B����ƿ����Һ��ɫ�ı仯
�ܵζ��ﵽ�յ�ʱ����Һ���ձ��
��3���ζ�������±���ʾ��
��NaOH����Һ��Ũ��Ϊ0.101 0mol/L�������Ʒ�е�����������Ϊ
������ȡ��Ʒ1.5g��
�������Ʒ�ܽ���250mL����ƿ�У����ݣ����ҡ�ȣ�
�������ȡ25.00mL��Ʒ��Һ��250mL��ƿ�У�����10mL 20%�����Լ�ȩ��Һ��ҡ�ȡ�����5min����1��2�η�̪��Һ����NaOH����Һ�ζ����յ㣮�����������������ظ�2�Σ�
��1����������������Ƿ���ȷ
��
��
����ǡ�����������ȷ�����������Ʒ�ܽ���С�ձ��У���ȴ����ת������ƿ
����Ʒ�ܽ���С�ձ��У���ȴ����ת������ƿ
��������ȷ���˿ղ����2�����ݲ������գ�
�ټ�ʽ�ζ���������ˮϴ�Ӻ�ֱ�Ӽ���NaOH����Һ���еζ���������Ʒ�е�����������
ƫ��
ƫ��
���ƫ�ߡ�����ƫ�͡�����Ӱ�족��������ƿ������ˮϴ�Ӻ�ˮδ��������ζ�ʱ��ȥNaOH����Һ�����
��Ӱ��
��Ӱ��
���ƫ����ƫС������Ӱ�족�����۵ζ�ʱ�ߵα�ҡ����ƿ���۾�Ӧ�۲�
B
B
��A���ζ�����Һ��ı仯
B����ƿ����Һ��ɫ�ı仯
�ܵζ��ﵽ�յ�ʱ����Һ���ձ��
��ɫ
��ɫ
ɫ����3���ζ�������±���ʾ��
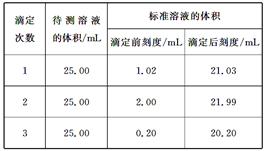
| �ζ� ���� |
������Һ�����/mL | ����Һ����� | |
| �ζ�ǰ�̶�/mL | �ζ���̶�/mL | ||
| 1 | 25.00 | 1.02 | 21.03 |
| 2 | 25.00 | 2.00 | 21.99 |
| 3 | 25.00 | 0.20 | 20.20 |
18.85%
18.85%
����������1������������ƿ���ܽ���壻
��2���ٵζ�����ҪҪNaOH��Һ��ϴ������ᵼ����ҺŨ��ƫ�ͣ����ƫ��
����ƿ���Ƿ���ˮ����ʵ������Ӱ�죬�ɴ����ʵ����ʵ����ĽǶȷ�����
�۵ζ�ʱ�۾�Ӧע��ע��۲���ɫ�仯����ȷ���յ㣻
�ܸ��ݷ�̪�ı�ɫ��Χȷ���ζ��յ�ʱ��ɫ�仯��
��3������Һ�����Ӧȡ����ʵ���ƽ��ֵ���������Һ��H+�����ʵ��������ݷ���ʽ��֪��CH2��6N4H+�����ʵ���������ȷ����Ʒ�е�������������
��2���ٵζ�����ҪҪNaOH��Һ��ϴ������ᵼ����ҺŨ��ƫ�ͣ����ƫ��
����ƿ���Ƿ���ˮ����ʵ������Ӱ�죬�ɴ����ʵ����ʵ����ĽǶȷ�����
�۵ζ�ʱ�۾�Ӧע��ע��۲���ɫ�仯����ȷ���յ㣻
�ܸ��ݷ�̪�ı�ɫ��Χȷ���ζ��յ�ʱ��ɫ�仯��
��3������Һ�����Ӧȡ����ʵ���ƽ��ֵ���������Һ��H+�����ʵ��������ݷ���ʽ��֪��CH2��6N4H+�����ʵ���������ȷ����Ʒ�е�������������
����⣺��1�������������ȷ������������ƿ���ܽ���壬����Ӧ���Ȱ���Ʒ�ܽ���С�ձ��У���ȴ����ת������ƿ���ʴ�Ϊ������Ʒ�ܽ���С�ձ��У���ȴ����ת������ƿ��
��2���ټ�ʽ�ζ���������ˮϴ�Ӻ���Ҫ����NaOH��Һ��ϴ�������൱��NaOH��Һ��ϡ�ͣ��ζ����ĵ������ƫ�ߣ������Ʒ�е�����������Ҳ��ƫ�ߣ�
�ʴ�Ϊ��ƫ�ߣ�
����ƿ������ˮϴ�Ӻ���Ȼˮδ������������Һ�е�H+�����ʵ������䣬��ζ�ʱ����NaOH����Һ�е��������Ƶ����ʵ����Ͳ��䣬Ҳ������Ӱ�죬
�ʴ�Ϊ����Ӱ�죻
�۶�ʱ�ߵα�ҡ����ƿ���۾�Ӧע��۲���ɫ�仯��ȷ���ζ��յ㣬�ʴ�Ϊ��B��
�ܴ���ҺΪ���ԣ���̪ӦΪ��ɫ������ҺתΪ����ʱ����Һ��ɫ��Ϊ�ۺ죨��dz�죩���ʴ�Ϊ��dz�죻
��3������Һ�����Ӧȡ����ʵ���ƽ��ֵ��
����ȷ���ζ�ʱ���õ�NaOH����ҺΪ
mL=20.00mL��
�����������Լ�ȩ��Һһ���ǹ����ģ�����1.500g ��� ���ܽ��ȡ������
���еζ�����0.15g��
�ζ��������Һ�к���H+������CH2��6N4H+����0.02L����0.1010mol/L=0.00202mol��
����4NH4++6HCHO�T3H++6H2O+��CH2��6N4H+��ÿ����4molH+������CH2��6N4H+����������NH4+4mol��
���Թ�����NH4+0.00202mol��
���к���Ԫ��0.00202mol��14g/mol=0.02828g
���Ե�����������Ϊ
��100%=18.85%��
�ʴ�Ϊ��18.85%��
��2���ټ�ʽ�ζ���������ˮϴ�Ӻ���Ҫ����NaOH��Һ��ϴ�������൱��NaOH��Һ��ϡ�ͣ��ζ����ĵ������ƫ�ߣ������Ʒ�е�����������Ҳ��ƫ�ߣ�
�ʴ�Ϊ��ƫ�ߣ�
����ƿ������ˮϴ�Ӻ���Ȼˮδ������������Һ�е�H+�����ʵ������䣬��ζ�ʱ����NaOH����Һ�е��������Ƶ����ʵ����Ͳ��䣬Ҳ������Ӱ�죬
�ʴ�Ϊ����Ӱ�죻
�۶�ʱ�ߵα�ҡ����ƿ���۾�Ӧע��۲���ɫ�仯��ȷ���ζ��յ㣬�ʴ�Ϊ��B��
�ܴ���ҺΪ���ԣ���̪ӦΪ��ɫ������ҺתΪ����ʱ����Һ��ɫ��Ϊ�ۺ죨��dz�죩���ʴ�Ϊ��dz�죻
��3������Һ�����Ӧȡ����ʵ���ƽ��ֵ��
����ȷ���ζ�ʱ���õ�NaOH����ҺΪ
| 20.01+19.99+20.00 |
| 3 |
�����������Լ�ȩ��Һһ���ǹ����ģ�����1.500g ��� ���ܽ��ȡ������
| 1 |
| 10 |
�ζ��������Һ�к���H+������CH2��6N4H+����0.02L����0.1010mol/L=0.00202mol��
����4NH4++6HCHO�T3H++6H2O+��CH2��6N4H+��ÿ����4molH+������CH2��6N4H+����������NH4+4mol��
���Թ�����NH4+0.00202mol��
���к���Ԫ��0.00202mol��14g/mol=0.02828g
���Ե�����������Ϊ
| 0.02828 |
| 0.15 |
�ʴ�Ϊ��18.85%��
���������⿼�����ʵĺ����IJⶨ���������к͵ζ��Ŀ��飬ע����ѧ��ʵ�������ͷ��������ͼ����������ۺϿ��飬Ϊ���Ը�Ƶ���㣬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
 һ����ʦ�����Ծ�ϵ�д�
һ����ʦ�����Ծ�ϵ�д� �����Ծ���Ԫ���Ծ�ϵ�д�
�����Ծ���Ԫ���Ծ�ϵ�д�
�����Ŀ