��Ŀ����
����Ŀ����������( NaCl02)��һ�ָ�Ч��������Ư������Ҫ�����ġ�ֽ��Ư�ס�ʳƷ������ˮ�����ȡ���֪��NaClO2������Һ���¶ȵ���38 ��ʱ�����ľ�����NaClO2![]() 3H2O������38 ��ʱ����������NaClO2������60��ʱNaClO2�ֽ��NaClO3��NaCl����ClO2�ֽⱬը��һ���Ʊ��������ƴֲ�Ʒ�Ĺ����������£�
3H2O������38 ��ʱ����������NaClO2������60��ʱNaClO2�ֽ��NaClO3��NaCl����ClO2�ֽⱬը��һ���Ʊ��������ƴֲ�Ʒ�Ĺ����������£�
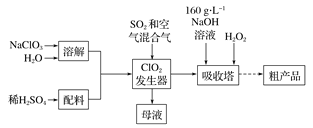
��1�� ClO2�������е����ӷ���ʽΪ ���������й��˿��������ÿ�����__________��ѡ����ţ���
a����SO2������SO3����ǿ����
b��ϡ��ClO2�Է�ֹ��ը
c����NaClO3��ԭΪClO2
��2���������ڷ�Ӧ�Ļ�ѧ����ʽΪ �����������¶Ȳ��ܳ���20������ԭ����_____________��
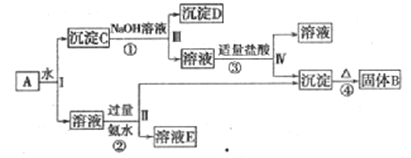
��3������ĸҺ���пɻ��յ���Ҫ������ ��
��4�����������пɻ��NaCl02��Һ����NaCl02��Һ���ֲ�Ʒ(NaClO2)�����IJ�����������Ϊ������ѹ��55�������ᾧ���� ���� ��������60������õ���Ʒ��
��5��Ϊ�ⶨ��Ʒ��NaCl02������������������ʵ�飺
ȷ��ȡ��������������Ʒ10.00 g���ձ��У�������������ˮ�����ĵ⻯�ؾ��壬�ٵ���������ϡ���ᣬ��ַ�Ӧ��ClO2-+ 4I-+ 4H+= 2H2O+ 2I2+ Cl-���������û��Һ���250mL������Һ��ȡ25.00 mL����Һ����2.000 mol��L-lNa2S203��Һ�ζ���I2+2S2O32-= 2I-+S4O62-�����������Na2SO3��Һƽ��ֵΪ16.40mL������Ʒ��NaClO2����������Ϊ ��
���𰸡���1��2ClO3-��SO2��2ClO2����SO42- b��
��2��2NaOH+2ClO2+H2O2=2NaClO2+2H2O+O2������ֹH2O2�ֽ�
��3��Na2SO4��4�����ˣ�38-60����ˮϴ�ӣ�5��74.21%
�������������������1��ClO2�������������ơ�����������������ԭ��Ӧ���ɶ������ȣ����ӷ���ʽΪ2ClO3-��SO2��2ClO2����SO42-����ClO2�ֽⱬը���������й��˿�����������ϡ��ClO2�Է�ֹ��ը��
��2����������ClO2��������⡢�������Ʒ�Ӧ�����������ƵĻ�ѧ����ʽΪ2NaOH + 2ClO2 + H2O2 = 2NaClO2 + 2H2O + O2����Ϊ��ֹH2O2�ֽ⣬���������¶Ȳ��ܳ���20����
��3��ClO2�������з�Ӧ���ɶ������ȡ������ƣ�����ĸҺ���пɻ��յ���Ҫ������Na2SO4��
��4�����������пɻ��NaCl O 2��Һ����NaCl O 2��Һ���ֲ�Ʒ(NaClO2)�����IJ�����������Ϊ������ѹ��55�������ᾧ�������ˣ���38-60����ˮϴ�ӣ�������60������õ���Ʒ��
��5������Ʒ��NaClO27.421g
����Ʒ��NaClO2����������Ϊ![]() 74.21%
74.21%

 ״Ԫ��ȫ��ͻ�Ƶ�����ϵ�д�
״Ԫ��ȫ��ͻ�Ƶ�����ϵ�д�