��Ŀ����
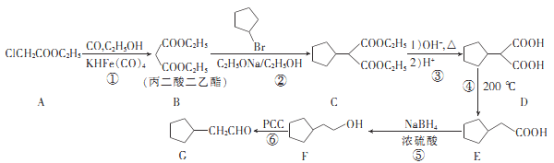
����Ŀ���������������ˮ����Ѫѹ��ҩ����м���G��һ�ֺϳ�·�����£�
�ش���������:
(1)A�Ļ�ѧ������__________��B�к��й����ŵ�����Ϊ___________��
(2)��Ӧ�ڵķ�Ӧ������____________��
(3)G������Cu(OH)2��Ӧ�Ļ�ѧ����ʽΪ__________________��
(4)X��E��Ϊͬ���칹�壬X�к�����Ԫ̼������X����NaOH��Һ��Ӧ�������������X�Ľṹ��ʽΪ___________________��
(5)�����1,3-�������ͱ�����������Ʊ�![]() �ĺϳ�·��(�����Լ���ѡ)��_____________
�ĺϳ�·��(�����Լ���ѡ)��_____________
���𰸡����������� ���� ȡ����Ӧ ![]() ��2Cu��OH��2��NaOH
��2Cu��OH��2��NaOH![]()
![]() ��
��![]() ��3H2O
��3H2O ![]() ��
��![]()
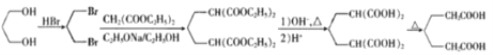
��������
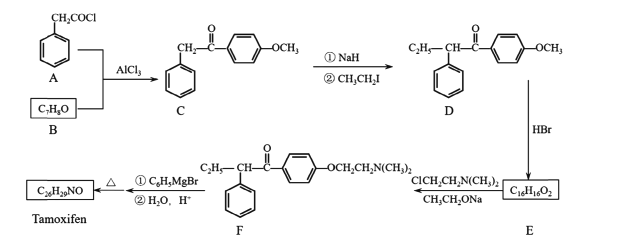
��ͼ��֪AΪ����������������������������±������ȡ����Ӧ�õ�B�������������B���屽����ȡ����Ӧ�õ�C��C����ˮ��õ�D��D��200��ʱ̼����һ���Ȼ����ѵõ�E��E����ԭ�õ�F���࣬F�����õ�Gȩ�࣬�ɴ˷�����
��1��A�����������Ҵ��γɵ�����A������Ϊ��������������B�Ľṹ��֪B�к��й�����Ϊ������
�ʴ�Ϊ��������������������
��2���Ա�B��C�Ľṹ�����C�ķ���ʽ����֪�������������һ�廷���鷢��ȡ����Ӧ����C��C��������ˮ�ⷴӦ����D��
�ʴ�Ϊ��ȡ����Ӧ��
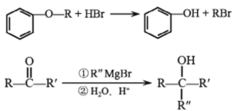
��3��G��ȩ��������Ϊ�Ȼ���ͬʱ��������ͭ��ˮ���Ȼ��ֿ�����NaOH�����кͷ�Ӧ����Ӧ����ʽΪ��![]() ��2Cu��OH��2��NaOH
��2Cu��OH��2��NaOH![]()
![]() ��
��![]() ��3H2O��
��3H2O��
�ʴ�Ϊ��![]() ��2Cu��OH��2��NaOH
��2Cu��OH��2��NaOH![]()
![]() ��
��![]() ��3H2O��
��3H2O��
��4��X��E��Ϊͬ���칹�壬X�к�����Ԫ̼������X����NaOH��Һ��Ӧ����X�����������Ȼ�����XΪ���ỷ���������ᣬX���ܽṹ��ʽΪ��![]() ��
��![]() ��
��
�ʴ�Ϊ��![]() ��
��![]() ��
��
��5��HOCH2CH2CH2OH��HBr����ȡ����Ӧ����BrCH2CH2CH2OH��Br��Ȼ�����������������Ҵ���/�Ҵ������³�![]() ������������ˮ����ữ����
������������ˮ����ữ����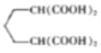 ����������������
����������������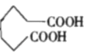 ���ϳ�·������ͼΪ��
���ϳ�·������ͼΪ��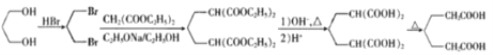 ��
��
�ʴ�Ϊ��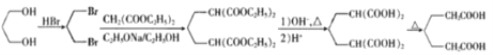 ��
��

����Ŀ����������ʵ���ó�����Ӧ������ȷ����
ʵ����ʵ | ���� | |
A | ����ͬ�¶��£���1 mL0.2 mol/LNaOH��Һ�е���2��0.1 mol/LMgCl2��Һ��������ɫ�������ٵμ�2��0.1 mol/LFeCl3��Һ�������ɺ��ɫ���� | �ܽ�ȣ�Mg(OH)2>Fe(OH)3 |
B | ij������ʹʪ�����ɫʯ����ֽ��� | ������ˮ��Һһ���Լ��� |
C | ͬ��ͬѹ�£������pH=3��HA��HB������ֱ���������п��Ӧ����ˮ���ռ����壬HA�ų����������ҷ�Ӧ���ʿ� | HB�����Ա�HAǿ |
D | SiO2����������ᷴӦ������Ӧ | SiO2������������ |
A.AB.BC.CD.D