��Ŀ����
X��Y��Z��Ϊ������Ԫ�أ�ԭ��������������X�ĵ���Ϊ�ܶ���С�����壬Yԭ������������������������������Z��Xԭ������������ͬ���ش��������⣺
��1��X��Y��Z��Ԫ�ط��ŷֱ�Ϊ �� �� ��
��2��������Ԫ����ɵĻ������У��Ⱥ��й��ۼ��ֺ������Ӽ����� �� ��
��3��X��Y��ɵĻ������У��Ⱥ��м��Թ��ۼ��ֺ��зǼ��Թ��ۼ����� ���˻������������������������ط�Ӧ�����ӷ���ʽΪ ���˻����ﻹ�ɽ����Թ�ҵ��ˮ�е�CN-����Ϊ̼���κͰ�����Ӧ�����ӷ���ʽΪ ��
ͨ������������������������ˮ�����ٷ�Һ�ŷŶԻ�������Ⱦ��ͬʱ����K2Cr2O7��
ʵ���ҶԺ�����Һ(����Cr3+��Fe3+��K+��SO42����NO3��������Cr2O72��)�����������ù������£�
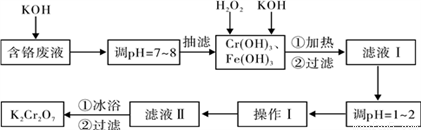
��֪����Cr(OH)3 + OH�� = CrO2�� + 2H2O��
��2CrO2�� + 3H2O2 + 2OH�� = 2CrO42�� + 4H2O��
��H2O2�����������¾��л�ԭ�ԣ��ܽ�+6��Cr��ԭΪ+3��Cr��
��1����ͼ����KOH��������250mL 6 mol��L��1 KOH��Һ�Ĺ���ʾ��ͼ��
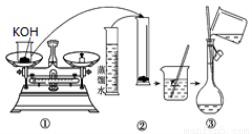
������۲�ͼʾ�жϣ����в���ȷ�IJ�����(�����)_____________________��
����������250 mL��Һ�����������(������)_________________��
�������ͼʾ�IJ���������Һ�������Ƶ���ҺŨ�Ƚ�________(�ƫ��ƫС��)��
(2)��Һ���ữǰ�����м��ȵ�Ŀ����_________________����ԡ�����˺�Ӧ��������ˮϴ��K2Cr2O7����Ŀ����_______��
��3���±���������ʵ��ܽ�����ݣ�
���� | 0�� | 20�� | 40�� | 60�� | 80�� | 100�� |
KCl | 28.0 | 34.2 | 40.1 | 45.8 | 51.3 | 56.3 |
K2SO4 | 7.4 | 11.1 | 14.8 | 18.2 | 21.4 | 24.1 |
K2Cr2O7 | 4.7 | 12.3 | 26.3 | 45.6 | 73.0 | 102.0 |
KNO3 | 13.9 | 31.6 | 61.3 | 106 | 167 | 246.0 |
�����ܽ�����ݣ�����������������Ϊ��________________��________________��
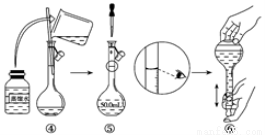
��ȡ��Ʒ�ظ��������2.000g���250mL��Һ��ȡ��25.00mL����ƿ�У�����10mL 2mol��L��1H2SO4�������⻯��(���Ļ�ԭ����ΪCr3+�������ڰ���5min��Ȼ�����100mLˮ������3mL����ָʾ������0.1200 mol��L��1Na2S2O3����Һ�ζ�(I2+2S2O32��=2I��+S4O62��)��
��д���ظ���������⻯�Ƶ����ӷ���ʽ___________________��
�ڵζ��յ������Ϊ___________________��
����ʵ���й���ȥNa2S2O3����Һ30.00mL�����ò�Ʒ�е��ظ���صĴ��� Ϊ_________(�������������������ʲ����뷴Ӧ)��
�����ζ�����ʹ��ǰδ��Na2S2O3����Һ��ϴ����õ��ظ���صĴ��Ƚ�_____________(�ƫ�ߡ�����ƫ�͡����䡱)��

