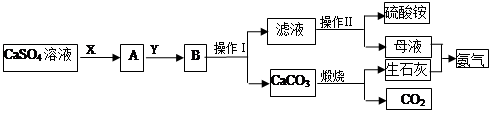��Ŀ����
4,7-�����㶹�أ��۵㣺132.6�棩��һ����Ҫ�����ϣ��㷺�ֲ���ֲ�����,�ɼ�ױ���Ϊԭ�ϵĺϳɷ�Ӧ���£�
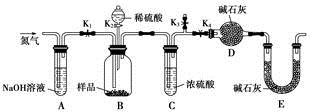
ʵ��װ��ͼ���£�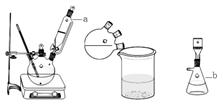
��Ҫʵ�鲽�裺
����1.��������ƿ�м���60mLŨ���ᣬ����ȴ��0�����£������µ����ױ���30mL(0.29mol)��������������26.4mL (0.21mol)�Ļ���
����2.������10���£�����12h����Ӧ��ȫ���䵹���ˮ������У�Ȼ����ˡ�ˮϴ�ô�Ʒ
����3.��Ʒ���Ҵ��ܽⲢ�ؽᾧ���ð�ɫ��״���岢��ɣ��Ƶò�Ʒ����Ϊ33.0g��
��1��ͼ����Ʒ���ƣ�a ��b ��
��2��ŨH2SO4��Ҫ��ȴ��0�����µ�ԭ���� ��
��3����Ӧ��Ҫ����12h����ԭ���� ��
��4��ȷ�����ղ�Ʒ��4,7-�����㶹�ص�ʵ����� ��
��5������ʵ�����Ϊ ��
��1����ѹ��Һ©����2�֣�������ƿ��2�֣���
��2����ֹŨ���Ὣ�л���������̿����2�֣���
��3��ʹ��Ӧ���ֽӴ���Ӧ����߷�Ӧ���ʣ�2�֣�/
��4�������۵��ⶨ����⣨���⣩���ף����˴Ź������ף��ȣ�2�֣���
��5��89.0%��2�֣���
���������������1����ѹ��Һ©��������ƿ����2��Ũ�������ǿ�����Ժ������ԣ��¶ȸ���ʹ�л�����������������������ˮ̼������3��ʹ��Ӧ���ֽӴ���Ӧ����߷�Ӧ���ʣ���4��4,7-�����㶹�أ��۵㣺132.6�棩�۵�ϵͣ����Բ����۵㣬Ҳ����ͨ��ʵ�������ⶨ����⣨���⣩���ף����˴Ź������ף��ȣ���5�����۲���Ϊ0.21��176=36.96g������Ϊ33.0��36.96=0.89
���㣺����ʵ�黯ѧ�������Ʊ�ԭ�����ʵ��й����⡣

�ӷϷ���������Ҫ�ɷ�V2O5��VOSO4��K2SO4��SiO2�ȣ��л���V2O5��һ��������������ʾ��ͼ���£���ش��������⣺
��1��������з�������Ҫ�ɷ��� ������X�Լ�Ϊ ��
��2��ʵ�����н�����ȡ��Һ����ʱ��ע����ȡ�������������Һ©������Ȧ�Ͼ��ã���Һ��ֲ�������IJ����� ��
��3���ڡ��۵ı仯���̿ɼ�Ϊ����ʽR��ʾVO2+��HA��ʾ�л���ȡ������
R2(SO4)n (ˮ��)+ 2nHA���л��㣩 2RAn���л��㣩 + nH2SO4 (ˮ��)Ϊ��ߢ�����ȡ�ٷ��ʣ�Ӧ��ȡ�Ĵ�ʩ�� ��
2RAn���л��㣩 + nH2SO4 (ˮ��)Ϊ��ߢ�����ȡ�ٷ��ʣ�Ӧ��ȡ�Ĵ�ʩ�� ��
��4������ɢ��еķ�Ӧ���ӷ���ʽ��
��ClO3- + ��VO2+ +��H+ =��VO3+ + �� +��
��5��25��ʱ��ȡ����������������õ��������ʺ���ҺpH֮���ϵ���±���
| pH | 1.3 | 1.4 | 1.5 | 1.6 | 1.7 | 1.8 | 1.9 | 2.0 | 2.1 |
| ��������% | 88.1 | 94.8 | 96.5 | 98.0 | 98.8 | 98.8 | 96.4 | 93.1 | 89.3 |
����ϱ�����ʵ�������У����м��백ˮ��������Һ�����pHֵΪ ��
��6���ù��������У�����ѭ�����õ������� ��
��14�֣�ij������;�㷺�������������Լ���ýȾ��������������ԭ�ϡ����ⶨ��������Ԫ�أ�Ħ������Ϊ482g/mol��Ϊ��һ��ȷ��������ɣ�ij��ѧ��ȤС����������ʵ�飺
��ȡ48.20g����������ˮ�����100mL��Һ��������Һ���ػ�ɫ��
��ȡ������Һ50mL���Թ��У�����������0.1mol/LNaOH��Һ�������ȣ����������徭�����ͨ��Ũ�����У�Ũ��������0.85g�������ĺ��ɫ�����������ˡ�ϴ�ӡ����պ��4.00g���塣
����ȡ������Һ50mL���Թ��У�����������BaCl2��Һ����������������İ�ɫ���� 23.30g��
��ش��������⣺
��1��ʵ����в�������ĵ���ʽ ��
��2�������ʵĻ�ѧʽΪ �������йظ����ʵ���;�������� ��
| A����Ѫ�� | B����ˮ�� | C�����ӷ�ˮ�ļ���Լ� | D������ |
��4����SO2����ͨ������ʵ���Һ�п��Թ۲쵽��ʵ�������� ��д���÷�Ӧ�����ӷ���ʽ ��
��5��Ϊ�˽�һ����֤����������Ԫ�صĻ��ϼۣ�ijͬѧ���������ʵ�鷽����ȡ�����������Թ��У���ˮ����ܽ⣬�μ����軯����Һ������Һ��Ѫ��ɫ������֤����������һ������Fe3+�������۸�ʵ������Ƿ���� ������������ߡ����������������� ��
��12�֣�����ѧ������ѧ�뼼����
�ظ�����ǹ�ҵ������ʵ���ҵ���Ҫ����������ҵ�ϳ��ø�������Ҫ�ɷ�ΪFeO��Cr2O3������ΪSiO2��Al2O3��Ϊԭ��������ʵ����ģ�ҵ���ø�������K2Cr2O7����Ҫ��������ͼ���漰����Ҫ��Ӧ��: 6FeO��Cr2O3+24NaOH+7KClO3=12Na2CrO4+3Fe2O3+7KCl+12H2O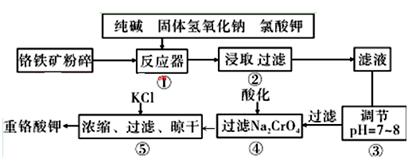
��1�����ǰ������������Ŀ���� ��
��2������۵���pH����˵õ��������� ��
��3���������У��ữʱ��CrO42-ת��ΪCr2O72-��д��ƽ��ת�������ӷ���ʽ ��
��4���ü�Ҫ������˵�������ݼ���KC1��ԭ��
��5��Ŀǰ��������Cr2O72-��ˮ������������巨���÷������ˮ�м���FeSO4 ��7H2O��Cr2O72-��ԭ��Cr3+������pH��Fe��Crת�����൱�� ����������,�������ֱ�ʾԪ�ؼ�̬���ij���������1mol Cr2O72-�������a mol FeSO4 ? 7H2O�����н�����ȷ����_______��
����������,�������ֱ�ʾԪ�ؼ�̬���ij���������1mol Cr2O72-�������a mol FeSO4 ? 7H2O�����н�����ȷ����_______��
| A��x ="0.5" ,a =8 | B��x ="0.5" ,a =" 10" | C��x =" 1.5" ,a =8 | D��x =" 1.5" ,a = 10 |
ij�о���ѧϰС��Ի�ԭ������ˮ�����ķ�Ӧ������п�ѧ̽������֪Ca(OH)2�ķֽ��¶�Ϊ580�棬������ˮ������Ӧ���¶�Ϊ900�棺������ͼ��ʾʵ��װ�ã������˻�ԭ������ˮ�����ķ�Ӧʵ�飬ʵ���й۲쵽����Һ�в����˴��������ݡ�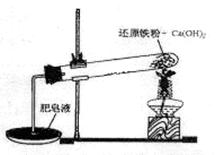
��1��ʵ����Ca(OH)2�������� ��ʵ���в�������������� ��
��2��Ϊ��һ��̽����ԭ������ˮ������Ӧ�������ijɷ֣��о���ѧϰС�齫��Ӧ��Ĺ��徭������õ���ɫ��������壬��Ժ�ɫ��������壬��С��������µļ��貢��������ص�ʵ�飺
����һ������ΪFeO
�����������ΪFe3O4
��������
����ѡ�������Լ������ᡢKSCN��Һ��K3Fe(CN)6 ��Һ����ˮ��֤������һ������
| ���� | ���� | ���� |
| | | ����һ������ |
��Ϊ�˽�һ��ȷ������ijɷ֣��ú�ɫ�������������ʵ��:

����������ˮ��������Ӧ�����ӷ���ʽ�� ��������ɫ�����Ƿ�ϴ�Ӹɾ��IJ����� ������ʱʢ�Ź���������� �����������������ɫ��������ȫ��ת���ɵĺ���ɫ��ĩ�������� g