��Ŀ����
( 14��)
(1)���������ڵ�����Ϊ���ʣ�����ȥ���и�������������������Լ���д�ں����ϣ�
��(�ױ�) ����(�Ҵ�) ���ױ�(��) ��
��2��ʵ�����ɵ�ʯ�е�̼���ƺ�ˮ��Ӧ��ȡ��Ȳ���÷�Ӧ�Ļ�ѧ����ʽ Ϊ ��ʵ����Ϊ�˼�����Ӧ���ʣ����� ����ˮ����ʵ���в���������������ŵ���ζ��������_________________�����Լ������Գ�ȥ��
Ϊ ��ʵ����Ϊ�˼�����Ӧ���ʣ����� ����ˮ����ʵ���в���������������ŵ���ζ��������_________________�����Լ������Գ�ȥ��
��3��ij��ȤС���ͬѧ��ʵ��������ȡ����ϩ�г������������� �����������������ʵ��ͼ��ȷ�����������������C2H4��SO2���ش��������⣺
�����������������ʵ��ͼ��ȷ�����������������C2H4��SO2���ش��������⣺
1��I��II��III��IVװ�ÿ�ʢ�ŵ��Լ�����Ϊ  ������ĸ��
������ĸ��
��Ʒ����Һ ��NaOH��Һ ��Ũ���� ������KMnO4��Һ
| A���ܢڢ٢� | B���٢ڢ٢� | C���٢ڢ٢� | D���ܢڢ٢� |
3��ʹ��װ��III��Ŀ���� ��
4��ȷ��������ϩ�������� ��
( 14��)
��1���������������Һ��ˮ�� NaOH��Һ��
��2��CaC2 + 2 H2O �� HCºCH��+ Ca(OH)2 ������ʳ��ˮ�� ����ͭ��Һ����NaOH��Һ��
��3�� 1��C 2��I��Ʒ����ɫ�� 3������SO2�Ƿ������ 4��IV��KMnO4��Һ��ɫ
����

(14��)�����£���ijһԪ�� ��
�� ��Һ�������ϣ����ǰ������Һ��Ũ�Ⱥͻ�Ϻ�������Һ��
��Һ�������ϣ����ǰ������Һ��Ũ�Ⱥͻ�Ϻ�������Һ�� ���±�(���Ϻ���Һ����仯���Բ���)��
���±�(���Ϻ���Һ����仯���Բ���)��
| ʵ���� |  ���ʵ���Ũ�� ���ʵ���Ũ���� 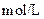 �� �� |  ���ʵ���Ũ�� ���ʵ���Ũ���� 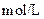 �� �� | �����Һ�� |
| �� |  |  | 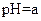 |
| �� |  | 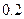 |  |
| �� | 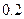 |  |  |
| �� | 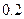 | 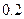 | 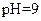 |
(1)�����ݼ������ݷ�����
 ��ȡֵ��ΧΪ ��
��ȡֵ��ΧΪ ��(2)���������������
 �Ƿ�һ������
�Ƿ�һ������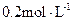 (ѡ��ǡ���)����
(ѡ��ǡ���)���� Ϊ���ᣬ������û����Һ��ƽ��ʱ
Ϊ���ᣬ������û����Һ��ƽ��ʱ ���볣��
���볣�� �Ľ���ֵ�� (�ú�
�Ľ���ֵ�� (�ú� ����ʽ��ʾ)��
����ʽ��ʾ)��(3)��������Һ
 ��ԭ���� ����������Һ������Ũ�ȵĴ�С��ϵ�� ��
��ԭ���� ����������Һ������Ũ�ȵĴ�С��ϵ�� ��(4)����ij����ˮ������ʵ���ռij���������ʵ����İٷ���������ˮ��ȡ������ݶ������ݣ���֪
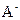 �ڸ������µ�ˮ����� ������Һ�У�ˮ�������
�ڸ������µ�ˮ����� ������Һ�У�ˮ�������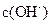 ��
��  ��
�� (14��) �����£���ijһԪ��HA��NaOH��Һ�������ϣ�������Һ��Ũ�Ⱥͻ�Ϻ�������Һ��pH���±���
|
ʵ���� |
HA���ʵ���Ũ�ȣ�mol/L�� |
NaOH���ʵ���Ũ�ȣ�mol/L�� |
��Ϻ���Һ��pH |
|
�� |
0.2 |
0.2 |
pH = a |
|
�� |
C1 |
0.2 |
pH = 7 |
|
�� |
0.2 |
0.1 |
pH >8 |
|
�� |
0.1 |
0.1 |
pH = 9 |
��ش�
��1���������������ʵ���������Ӽ�����������������a��˵��HA��ǿ�ỹ�����ᡣ
_______________________________________________________________________________��
��2���������������ʵ�����������������������C1�Ƿ�һ������0.2 mol/L________ �� ��Ϻ���Һ������Ũ��c(A-)��c(Na+)�Ĵ�С��ϵ��c(A-)_______ c(Na+) (��>��<��=)��
(3)�ӱ���ʵ����������HA��_______��(ǿ����)���û��Һ�е�����Ũ���ɴ�С��˳����______________________________________
��4��������Һ�У���ˮ�����c(H��) = mol��L-1��c (Na+)��c (A-)= mol��L-1��
��5����֪NH4A��ҺΪ���ԣ���֪��HA��Һ�ӵ�Na2CO3��Һ��������ų������ƶϣ�NH4��2CO3��Һ��pH 7 (��>��<��= )��
��6������ͬ�¶�����ͬŨ�ȵ���������Һ��
A��NH4HCO3 B��NH4A C��(NH4)2SO4 D��NH4Cl
��c(NH4+)�ɴ�С��˳������ ������ţ�