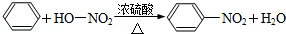
��Ŀ����
���п�ͼ��ʾ��ת����ϵ�У�A��C��Ϊ�����Ľ������ʣ� EΪ����ǽ������ʡ�BΪ�غ�ɫ���������XΪ��������ɫҺ�塣L��ɫΪ��ɫ������ʹ��̪��죨��Ӧ���������ɵ�ˮ��������������ȥ��

��ش��������⣺
��1��F�Ļ�ѧʽΪ ��
��2��I����ˮ��Ӧ�����ӷ���ʽΪ ��
��3��A��B����C��D�ķ�Ӧ�� ����ų��������ա������������ķ�Ӧ��
��4��D��L��Ӧ�����ӷ���ʽΪ ��
��5�����顰��ɫ��Һ���н��������ӵ��Լ��� ��д��ѧʽ��
 ��ְٷְټ���ϵ�д�
��ְٷְټ���ϵ�д� �����ƻ���ĩ��̶�100��ϵ�д�
�����ƻ���ĩ��̶�100��ϵ�д� �ܿ���ȫ��100��ϵ�д�
�ܿ���ȫ��100��ϵ�д��Ȼ�ͭ���壨CuCl2��2H2O���к���FeCl2���ʣ�Ϊ�Ƶô������Ȼ�ͭ���壬���Ƚ����Ƴ�ˮ��Һ���ٰ���ͼ��ʾ�IJ�����������ᴿ��

������±�������������ա�
�������↑ʼ����ʱ��pH | �������������ȫʱ��pH | |
Cu2�� | 4.7 | 6.7 |
Fe2�� | 7.0 | 9.0 |
Fe3�� | 1.9 | 3.7 |
��1������I����X��Ŀ����______________________��
��2���������ʶ�������ΪX���ʺϱ�ʵ���X���ʿ����ǣ���ѡ�_______��
A��KMnO4 B��NaClO C��H2O2 D��Cl2
��3������II��Y�Լ�Ϊ____________���ѧʽ������������Һ��pH��______��Χ��
��4��ʵ����������CuCl2��Һʱ���������_________�����������ƣ��������ǣ��������������й����ӷ���ʽ��ʾ��____________________��
��5������CuCl2��Һ�������գ��õ��Ĺ�����_________________���ѧʽ������Ҫ����ˮCuCl2���������������________________________________��


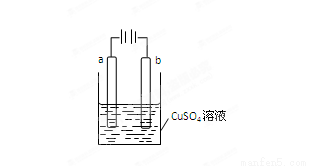
 bΪ����ͨ���Cu2+ ��a�缫�ƶ�
bΪ����ͨ���Cu2+ ��a�缫�ƶ�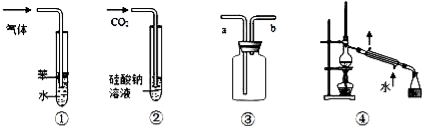
 ��������
�������� CH3Cl+HCl
CH3Cl+HCl 2CH3CHO+2H2O
2CH3CHO+2H2O CH3COOC2H5+H2O
CH3COOC2H5+H2O