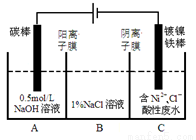
��Ŀ����
������ԭ�ζ�ʵ�����к͵ζ����ƣ�������֪Ũ�ȵ���������Һ�ζ�δ֪Ũ�ȵĻ�ԭ����Һ��֮������0. 00lmol/L����KMnO4��Һ�ζ�δ֪Ũ�ȵ�NaHSO3��Һ����ش��������⣺
(1)��ɵζ������ӷ���ʽ����__����ϵ������___�������ʻ����ӣ���_____
____MnO4-+____HSO3-+____=____Mn2++___ ____+___H2O
(2)�õζ�ʵ���������������̨���ζ��ܼ��⣬����Ҫ������Ʒ�е�_____������ţ�
A.��ʽ�ζ��� B.��ʽ�ζ��� C.©�� D.��ƿ E.�ձ� F.��ֽ
(3) KMnO4��Һ������____���ᡢ�ʽ�ζ���ʢװ��ԭ���ǣ�_______��
(4)�Ƿ���Ҫѡ��ָʾ����_____���ζ��յ��������___________��
(5)�ζ�ǰƽ��KMnO4Һ�棬�̶�ΪamL���ζ�����Һ���ض�ΪbmL����(b-a)mL��ʵ������KMnO4��Һ���_______�������С������Ӱ�족����ͬ��������(b-a) mL���㣬�õ�NaHSO3��Ũ�ȱ�ʵ�ʵ�Ũ��_______��
 ��У����ϵ�д�
��У����ϵ�д���֪H2(g)+Br2(l)=2HBr(g);��H=��72KJ/mol������1molBr2(l)��Ҫ���յ�����Ϊ30KJ������������������±���
H2(g) | Br2(g) | HBr(g) | |
1mol�����л�ѧ������ ʱ��Ҫ���յ�����/kJ | 436 | a | 369 |
�����aΪ
A. 404 B. 260 C. 230 D. 200


 B+HCOOH������ƽ������ϸ÷�Ӧ�������л���B�У����������칹��
B+HCOOH������ƽ������ϸ÷�Ӧ�������л���B�У����������칹��