��Ŀ����
�����8�֣�ij�о�С����2 L�ܱ������м�������Ļ���̿��0.2 mol NO������(T1��)�·�����Ӧ��C(s)��2NO(g) N2(g)��CO2(g)��Q(Q>0)��30 min��ﵽƽ�⣬���NOŨ��Ϊ0.04 mol/L���ش��������⣺
N2(g)��CO2(g)��Q(Q>0)��30 min��ﵽƽ�⣬���NOŨ��Ϊ0.04 mol/L���ش��������⣺
1���÷�Ӧ��ƽ�ⳣ������ʽK��______��T1��ﵽƽ��ʱ��N2��ƽ����Ӧ���ʣ�_______��
2��30 min�����ı�ijһ�������NO��ת���ʣ�����Ըı��������______________��
3��30 min�������¶���T2�棬�ﵽƽ���������NO��N2��CO2�Ĺ�ϵ��������____��
a��5:3:3 b��1:1:1 c��4:3:3 d��2:1:1
4������ʼʱ�ܱ����������Ϊ1 L�������������䣬�ﵽƽ�����ԭƽ����ȣ�����˵����ȷ����_________��
a��NO��ת���ʲ��� b��N2��Ũ����ԭ����2��
c����Ӧ�ų�������Ϊ0.1Q d���ﵽƽ���ʱ����ԭ����һ��
 N2(g)��CO2(g)��Q(Q>0)��30 min��ﵽƽ�⣬���NOŨ��Ϊ0.04 mol/L���ش��������⣺
N2(g)��CO2(g)��Q(Q>0)��30 min��ﵽƽ�⣬���NOŨ��Ϊ0.04 mol/L���ش��������⣺1���÷�Ӧ��ƽ�ⳣ������ʽK��______��T1��ﵽƽ��ʱ��N2��ƽ����Ӧ���ʣ�_______��
2��30 min�����ı�ijһ�������NO��ת���ʣ�����Ըı��������______________��
3��30 min�������¶���T2�棬�ﵽƽ���������NO��N2��CO2�Ĺ�ϵ��������____��
a��5:3:3 b��1:1:1 c��4:3:3 d��2:1:1
4������ʼʱ�ܱ����������Ϊ1 L�������������䣬�ﵽƽ�����ԭƽ����ȣ�����˵����ȷ����_________��
a��NO��ת���ʲ��� b��N2��Ũ����ԭ����2��
c����Ӧ�ų�������Ϊ0.1Q d���ﵽƽ���ʱ����ԭ����һ��
�����8�֣�ÿС��2�֣�
1��K= ��0.001 mol/(L?min)
��0.001 mol/(L?min)
2�����¡���ȥ������̼��2�֣���
3��b��c��2�֣���
4��a��b��2�֣�
1��K=
 ��0.001 mol/(L?min)
��0.001 mol/(L?min)2�����¡���ȥ������̼��2�֣���
3��b��c��2�֣���
4��a��b��2�֣�
1��C(s)��2NO(g) N2(g) �� CO2(g)
N2(g) �� CO2(g)
��ʼ�� 0.2mol 0 0
��n 0.12mol 0.06mol 0.06mol
30 min: 0.08mol 0.06mol 0.06mol
3��30 minƽ��ʱ����������ʵ�����Ϊ��4:3:3��������ƽ�����ƣ�NO���࣬�����������
4������ʼʱ�ܱ����������Ϊ1 L��������Ϊ��ѹ��ƽ�ⲻ�ƶ������������Ũ�ȵȱ������ӡ�
 N2(g) �� CO2(g)
N2(g) �� CO2(g)��ʼ�� 0.2mol 0 0
��n 0.12mol 0.06mol 0.06mol
30 min: 0.08mol 0.06mol 0.06mol
3��30 minƽ��ʱ����������ʵ�����Ϊ��4:3:3��������ƽ�����ƣ�NO���࣬�����������
4������ʼʱ�ܱ����������Ϊ1 L��������Ϊ��ѹ��ƽ�ⲻ�ƶ������������Ũ�ȵȱ������ӡ�

��ϰ��ϵ�д�
 A�ӽ��� ϵ�д�
A�ӽ��� ϵ�д� ȫ�Ų��Ծ�ϵ�д�
ȫ�Ų��Ծ�ϵ�д�
�����Ŀ
 C(g)+D(g)���������������������仯ʱ���ܱ����÷�Ӧ�Ѵﵽ�������µ�����ȵ���
C(g)+D(g)���������������������仯ʱ���ܱ����÷�Ӧ�Ѵﵽ�������µ�����ȵ���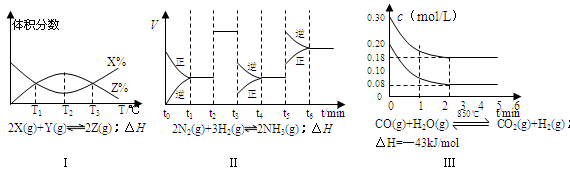
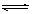 2C(g)���ﵽƽ��ı�־��
2C(g)���ﵽƽ��ı�־��  PCl3��g��+Cl2��g������H1>0 K1=1mol��L��1
PCl3��g��+Cl2��g������H1>0 K1=1mol��L��1 bM��g����M��N�����ʵ�����ʱ��ı仯������ͼ��ʾ��
bM��g����M��N�����ʵ�����ʱ��ı仯������ͼ��ʾ��
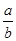 =__________
=__________  PCl3��g����Cl2��g������ʱѹǿΪP1������������ͨ��amol PCl5����ͬ�����ִﵽƽ��ʱ��ѹǿΪP2��������˵������ȷ���� �� ��
PCl3��g����Cl2��g������ʱѹǿΪP1������������ͨ��amol PCl5����ͬ�����ִﵽƽ��ʱ��ѹǿΪP2��������˵������ȷ���� �� �� 2CrO42����2H������K2Cr2O7����ˮ���ϡ��Һ, ��Һ�ʳ�ɫ��
2CrO42����2H������K2Cr2O7����ˮ���ϡ��Һ, ��Һ�ʳ�ɫ��