��Ŀ����
A��B��C��D��E���ֶ�����Ԫ�ص�ԭ����������������֪��
(1)��ԭ�Ӱ뾶��С��ϵ�ǣ�D>E>B>C>A��
(2)A��Dͬ���壬�����γ����Ӿ���DA��
(3)B��D��E���ߵ�����������Ӧ��ˮ��������֮����ɷ�Ӧ���ɿ������κ�ˮ�����������о���CԪ�ء�
(4)B��E����Ԫ��ԭ�ӵ�����������֮�͵���A��C��D����Ԫ��ԭ�ӵ�����������֮�͡�
����д���пհף�
(1)DA��ˮ��Ӧ��������Һ��pH_________(�>����<����=��)7��
(2)D2C2�ĵ���ʽ__________________��
(3)1 mol E������������D������������ˮ������Һ��Ӧ���ܲ���_________mol A2���壬��д���䷴Ӧ�����ӷ���ʽ__________________��
(4)д��B��D��E���ߵ�����������Ӧ��ˮ��������֮����ɷ�Ӧ���ɿ������κ�ˮ�����ӷ���ʽ��
_____________________________________________________��
_____________________________________________________��
_____________________________________________________��
_____________________________________________________��
(5)������BA3��BC�ڼ��Ⱥ��д������ڵ��������ܷ�����Ӧ���������������ʣ��䷴Ӧ�Ļ�ѧ����ʽΪ___________________________��
(1)>
(2)![]()
(3)![]() 2Al+2OH-+2H2O====2
2Al+2OH-+2H2O====2![]() +3H2��
+3H2��
(4)H++OH-====H2O
Al(OH)3+3H+====Al3++3H2O
Al(OH)3+OH-====![]() +2H2O
+2H2O
(5)4NH3+6NO ![]() 5N2+6H2O
5N2+6H2O
����:
���⿼��Ԫ�ؼ��仯������й����ʣ�����֪ʶ���ۺ�����������������A��Dͬ���壬A��ԭ��������ԭ�Ӱ뾶��С��D��ԭ�Ӱ뾶���֪��AΪH��DΪNa�����õ����о�����CԪ�أ���֪CԪ��Ϊ��Ԫ�أ���(4)��֪BΪN�� EΪAl��NaH��ˮ��Ӧ����NaOH��H2����Һ�Լ��ԡ�Na2O2�Ǻ��й����������ӻ����HNO3��NaOH��Al(OH)3����֮��������Ӧ������������кͺ�Al(OH)3�����ԡ�NH3��NO����Ӧ����N2��ˮ��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�


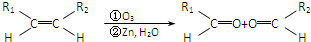

 Ԫ�����ڱ������ڵ�һ���������ʾ�������й�A��B��C��D��E����Ԫ�ص������У���ȷ���ǣ�������
Ԫ�����ڱ������ڵ�һ���������ʾ�������й�A��B��C��D��E����Ԫ�ص������У���ȷ���ǣ�������