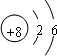��Ŀ����
��A��B��C��D��E��F����Ԫ�أ���֪��
������λ��������ͬ�����ڣ��˵������������
��EԪ�صĵ��������ݼ��±���kJ?mol-1����
��B��F�ֱ�ͬ���壮
��A��E�ֱ�����D��ԭ�Ӹ�����1�s1��2�sl�γɻ����
��B��C�ֱ�����D��ԭ�Ӹ�����1�s1��1�s2�γɻ����
��1��д��ֻ����A��B��D��E����Ԫ�ص��εĻ�ѧʽ
��2��B2A2�������
��3���±��г�����������Ԫ���γɵĻ�ѧ���ļ��ܣ�
���������������γɵľ��壬�۵��ɸߵ��͵�˳����a��b��c��d��ʾ����
a��F��D�γɵĻ����� b��B��D�γɵ��ȶ������� c��F�ĵ��� d��E��D�γɵĻ�����
����д��1molF���ʾ����������Ӧ���Ȼ�ѧ����ʽ
������λ��������ͬ�����ڣ��˵������������
��EԪ�صĵ��������ݼ��±���kJ?mol-1����
| I1 | I2 | I3 | I4 | �� |
| 496 | 4562 | 6912 | 9540 | �� |
��A��E�ֱ�����D��ԭ�Ӹ�����1�s1��2�sl�γɻ����
��B��C�ֱ�����D��ԭ�Ӹ�����1�s1��1�s2�γɻ����
��1��д��ֻ����A��B��D��E����Ԫ�ص��εĻ�ѧʽ
NaHCO3
NaHCO3
����2��B2A2�������
3
3
���Ҽ���2
2
���м�����3���±��г�����������Ԫ���γɵĻ�ѧ���ļ��ܣ�
| ��ѧ�� | F-D | F-F | B-B | F-B | C�TD | D�TD |
| ����/kJ?mol-1 | 460 | 176 | 347.7 | 347 | 745 | 497.3 |
a��c��d��b
a��c��d��b
��a��F��D�γɵĻ����� b��B��D�γɵ��ȶ������� c��F�ĵ��� d��E��D�γɵĻ�����
����д��1molF���ʾ����������Ӧ���Ȼ�ѧ����ʽ
Si��s��+O2��g��=SiO2��s����H=-989.2kJ/mol
Si��s��+O2��g��=SiO2��s����H=-989.2kJ/mol
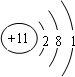
����������E�ĵ����ܿ�֪�����Ӧ��1�����ӣ�ӦΪNaԪ�أ�A��E�ֱ�����D��ԭ�Ӹ�����1�s1��2�sl�γɻ������֪EΪNa����DӦΪO�����γ�Na2O2��Na2O��AΪHԪ�أ�����O�γ�H2O2��H2O��B��C�ֱ�����D��ԭ�Ӹ�����1�s1��1�s2�γɻ������֪DΪO����B��C����ΪC��NԪ�أ���˵��������������BΪCԪ�أ�CΪNԪ�أ�B��F�ֱ�ͬ���壬��FΪSi����϶�Ӧ�����ʵ����ʺ���ĿҪ������⣮
����⣺��E�ĵ����ܿ�֪�����Ӧ��1�����ӣ�ӦΪNaԪ�أ�A��E�ֱ�����D��ԭ�Ӹ�����1�s1��2�sl�γɻ������֪EΪNa����DӦΪO�����γ�Na2O2��Na2O��AΪHԪ�أ�����O�γ�H2O2��H2O��B��C�ֱ�����D��ԭ�Ӹ�����1�s1��1�s2�γɻ������֪DΪO����B��C����ΪC��NԪ�أ���˵��������������BΪCԪ�أ�CΪNԪ�أ�B��F�ֱ�ͬ���壬��FΪSi��
��1��ֻ����A��B��D��E����Ԫ�ص���ΪNaHCO3���ʴ�Ϊ��NaHCO3��
��2��B2A2ΪC2A2���ṹʽΪH-C��C-H������3���Ҽ���2���м����ʴ�Ϊ��3��2��
��3����a��F��D�γɵĻ�����ΪSiO2��
b��B��D�γɵ��ȶ�������ΪCO2��
c��F�ĵ���ΪSi��
d��E��D�γɵĻ�����ΪNa2O2��Na2O��
����SiO2��SiΪԭ�Ӿ��壬�е�ϸߣ���SiO2��Si��Na2O2��Na2OΪ���Ӿ��壬CO2Ϊ���Ӿ��壬�۵��ɸߵ��͵�˳��a��c��d��b��
�ʴ�Ϊ��a��c��d��b��
��1molSi�����к���2molSi-Si������������Ӧ�Ļ�ѧ����ʽΪSi+O2=SiO2����Ӧ�ȵ��ڷ�Ӧ����ܼ��ܼ���������ܼ��ܣ�����1molSiO2����4mol Si-O�
���H=2��176kJ/mol+497.3kJ/mol-4��460kJ/mol=-989.2kJ/mol��
�ʴ�Ϊ��Si��s��+O2��g��=SiO2��s����H=-989.2kJ/mol��
��1��ֻ����A��B��D��E����Ԫ�ص���ΪNaHCO3���ʴ�Ϊ��NaHCO3��
��2��B2A2ΪC2A2���ṹʽΪH-C��C-H������3���Ҽ���2���м����ʴ�Ϊ��3��2��
��3����a��F��D�γɵĻ�����ΪSiO2��
b��B��D�γɵ��ȶ�������ΪCO2��
c��F�ĵ���ΪSi��
d��E��D�γɵĻ�����ΪNa2O2��Na2O��
����SiO2��SiΪԭ�Ӿ��壬�е�ϸߣ���SiO2��Si��Na2O2��Na2OΪ���Ӿ��壬CO2Ϊ���Ӿ��壬�۵��ɸߵ��͵�˳��a��c��d��b��
�ʴ�Ϊ��a��c��d��b��
��1molSi�����к���2molSi-Si������������Ӧ�Ļ�ѧ����ʽΪSi+O2=SiO2����Ӧ�ȵ��ڷ�Ӧ����ܼ��ܼ���������ܼ��ܣ�����1molSiO2����4mol Si-O�
���H=2��176kJ/mol+497.3kJ/mol-4��460kJ/mol=-989.2kJ/mol��
�ʴ�Ϊ��Si��s��+O2��g��=SiO2��s����H=-989.2kJ/mol��
�����������ۺϿ���Ԫ��λ�á��ṹ�����ʵĹ�ϵ���ۺϿ���ѧ���ķ����������ƶ��������ۺ����û�ѧ֪ʶ���������ѶȽϴ�ע������Ȼ�ѧ����ʽ����д��

��ϰ��ϵ�д�
 ֥�鿪���γ�������ϵ�д�
֥�鿪���γ�������ϵ�д� ����ѧ��ţ��Ӣ��ϵ�д�
����ѧ��ţ��Ӣ��ϵ�д�
�����Ŀ