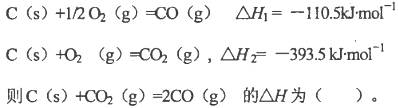��Ŀ����
ij�жԴ������м�⣬���ָ�����Ҫ��Ⱦ��Ϊ�����������PM2.5��ֱ��С�ڵ���2.5 �����������������Ҫ��ԴΪȼú��������β���ȡ���ˣ���PM2.5��SO2��NOx�Ƚ����о�������Ҫ���塣
�����������������Ҫ��ԴΪȼú��������β���ȡ���ˣ���PM2.5��SO2��NOx�Ƚ����о�������Ҫ���塣
��ش��������⣺
��1����PM2.5����������ˮ�����Ƴɴ���������
����ø������������ӵĻ�ѧ��ּ���Ũ�����±���
���ݱ��������ж�������pH= ��
��2��Ϊ����SO2���ŷţ�����ȡ�Ĵ�ʩ�У�
�ٽ�úת��Ϊ�������ȼ�ϡ�
��֪��H2��g��+1/2O2��g��=H2O��g�� ��H=��241.8kJ��mol��1
C��s��+1/2O2��g��="CO" ��g�� ��H=��110.5kJ��mol��1
д����̿��ˮ������Ӧ���Ȼ�ѧ����ʽ�� ���� ��
��ϴ�Ӻ�SO2���������������ʿ���ϴ�Ӽ����� ��
A��Ca(OH) 2 B��Na2CO3 C��CaCl2D��NaHSO3
��3������β������NOx��CO�����ɼ�ת��
�� ��1mol������0.8molN2��0.2molO2�������еĻ�ѧ��ӦʽΪN2 (g)+O2(g) 2NO(g) ��H
2NO(g) ��H 0
0
1300��ʱ��1mol���������ܱ������ڷ�Ӧ�ﵽƽ�⣬���NOΪ8��10��4mol��������¶��µ�ƽ�ⳣ��K= ��
���������������¶�Խ�ߣ���λʱ����NO�ŷ���Խ����ԭ���� ��
��Ŀǰ��������β��ϵͳ��װ�ô�ת�����ɼ���CO��NOx����Ⱦ���仯ѧ��Ӧ����ʽΪ ���������������������������������������� ��
�� ����ȼ�Ͳ���ȫȼ��ʱ����CO���������밴���з�Ӧ��ȥCO��2CO��g��=2C��s��+O2��g��
��֪�÷�Ӧ�ġ�H 0���жϸ������ܷ�ʵ�ֲ����������ݣ� ��
0���жϸ������ܷ�ʵ�ֲ����������ݣ� ��
 �����������������Ҫ��ԴΪȼú��������β���ȡ���ˣ���PM2.5��SO2��NOx�Ƚ����о�������Ҫ���塣
�����������������Ҫ��ԴΪȼú��������β���ȡ���ˣ���PM2.5��SO2��NOx�Ƚ����о�������Ҫ���塣��ش��������⣺
��1����PM2.5����������ˮ�����Ƴɴ���������
����ø������������ӵĻ�ѧ��ּ���Ũ�����±���
| ���� | H+ | K+ | Na+ | NH4+ | SO42�� | NO3�� | Cl�� |
| Ũ��/mol��L��1 | δ�ⶨ | 4��10��6 | 6��10��6 | 2��10��5 | 4��10��5 | 3��10��5 | 2��10��5 |
���ݱ��������ж�������pH= ��
��2��Ϊ����SO2���ŷţ�����ȡ�Ĵ�ʩ�У�
�ٽ�úת��Ϊ�������ȼ�ϡ�
��֪��H2��g��+1/2O2��g��=H2O��g�� ��H=��241.8kJ��mol��1
C��s��+1/2O2��g��="CO" ��g�� ��H=��110.5kJ��mol��1
д����̿��ˮ������Ӧ���Ȼ�ѧ����ʽ�� ���� ��
��ϴ�Ӻ�SO2���������������ʿ���ϴ�Ӽ����� ��
A��Ca(OH) 2 B��Na2CO3 C��CaCl2D��NaHSO3
��3������β������NOx��CO�����ɼ�ת��
�� ��1mol������0.8molN2��0.2molO2�������еĻ�ѧ��ӦʽΪN2 (g)+O2(g)
 2NO(g) ��H
2NO(g) ��H 0
01300��ʱ��1mol���������ܱ������ڷ�Ӧ�ﵽƽ�⣬���NOΪ8��10��4mol��������¶��µ�ƽ�ⳣ��K= ��
���������������¶�Խ�ߣ���λʱ����NO�ŷ���Խ����ԭ���� ��
��Ŀǰ��������β��ϵͳ��װ�ô�ת�����ɼ���CO��NOx����Ⱦ���仯ѧ��Ӧ����ʽΪ ���������������������������������������� ��
�� ����ȼ�Ͳ���ȫȼ��ʱ����CO���������밴���з�Ӧ��ȥCO��2CO��g��=2C��s��+O2��g��
��֪�÷�Ӧ�ġ�H
 0���жϸ������ܷ�ʵ�ֲ����������ݣ� ��
0���жϸ������ܷ�ʵ�ֲ����������ݣ� ����1��PH=4
��2����C(s)+H2O(g)=H2(g)+CO(g) ��H=+131.3kJ/mol �� A B
��3����4��10-6���¶�Խ�ߣ���Ӧ����Խ�� ��2XCO+2NOX 2XCO2+N2
2XCO2+N2
�۲���ʵ�֣���Ϊ�÷�Ӧ�ġ�H>0,��S<0,���ԡ�G>0
��2����C(s)+H2O(g)=H2(g)+CO(g) ��H=+131.3kJ/mol �� A B
��3����4��10-6���¶�Խ�ߣ���Ӧ����Խ�� ��2XCO+2NOX
 2XCO2+N2
2XCO2+N2�۲���ʵ�֣���Ϊ�÷�Ӧ�ġ�H>0,��S<0,���ԡ�G>0
�����������1��������Һ�ʵ����Ե�ԭ���ɵ�c(H+)+c(K+)+c(Na+)+c(NH4+)=2c(SO42��)+c(NO3��)+c(Cl��)����������ֵ��������ʽ�ӿɵ�c(H+)=1.0��10��4mol/L,����pH=4����2���� �ڣ��������ɵ�C(s)+H2O(g)=H2(g)+CO(g) ��H=+131.3kJ/mol����A��Ca(OH) 2+ SO2=CaSO3��+H2O����ȷ��B��Na2CO3+ SO2=Na2SO3+ CO2����ȷ��C��CaCl2����Ӧ�����������ռ�������D��NaHSO3����Ӧ�����������ռ�������3�����ڷ�Ӧ��ʼʱ��n(N2)=0.8mol��n(O2)=0.2mol;n(NO)=0;����Ӧ�ﵽƽ��ʱ��n(N2)=(0.8-4��10-4)mol
n(O2)=(0.2-4��10-4)mol; n(NO)= 8��10-4mol.���������ݻ�ΪVL������¶��µ�ƽ�ⳣ��
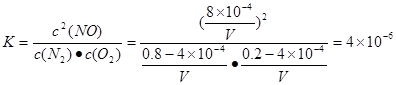 �����������������¶�Խ�ߣ���λʱ����NO�ŷ���Խ������Ϊ�����¶ȣ���ѧ��Ӧ���ʼӿ졣�ڸ�����֪������������غ㶨�ɿɵ÷�Ӧ����ʽΪ��2XCO+2NOX
�����������������¶�Խ�ߣ���λʱ����NO�ŷ���Խ������Ϊ�����¶ȣ���ѧ��Ӧ���ʼӿ졣�ڸ�����֪������������غ㶨�ɿɵ÷�Ӧ����ʽΪ��2XCO+2NOX  2XCO2+N2�������ڷ�Ӧ2CO��g��=2C��s��+O2��g����H>0�Ǹ���ϵ���ҳ̶ȼ�С�����ȷ�Ӧ����H>0����S<0��������ϵ�������ܦ�G=��H-T����S>0��.���Ը����벻��ʵ�֡�
2XCO2+N2�������ڷ�Ӧ2CO��g��=2C��s��+O2��g����H>0�Ǹ���ϵ���ҳ̶ȼ�С�����ȷ�Ӧ����H>0����S<0��������ϵ�������ܦ�G=��H-T����S>0��.���Ը����벻��ʵ�֡�
��ϰ��ϵ�д�
�����Ŀ
 FeO(s)+CO(g) ��H=akJ��mol-1,ƽ�ⳣ��ΪK;
FeO(s)+CO(g) ��H=akJ��mol-1,ƽ�ⳣ��ΪK; CO2(g) ��H=bkJ��mol-1;
CO2(g) ��H=bkJ��mol-1;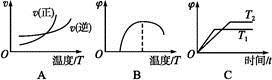
 2CO2(g)+N2(g)
2CO2(g)+N2(g)




 CH3OH(g)����ƽ��ʱ�����CO��H2��CH3OH�ֱ�Ϊ1 mol��1 mol��1 mol�����������Ϊ3L�����������м���ͨ��3 mol CO����ʱv������ v���棩��ѡ���>������<������=�������жϵ����� ��
CH3OH(g)����ƽ��ʱ�����CO��H2��CH3OH�ֱ�Ϊ1 mol��1 mol��1 mol�����������Ϊ3L�����������м���ͨ��3 mol CO����ʱv������ v���棩��ѡ���>������<������=�������жϵ����� �� CO(g)����H="-110.5" kJ��mol-1
CO(g)����H="-110.5" kJ��mol-1