��Ŀ����
����Ŀ������˵��ȫ����ȷ��һ����
��CH3��CH=CH2��CH2=CH2�����ʽ��ͬ ��CH��CH��C6H6��̼����ͬ
��̼ԭ������ͬ��ֱ������һ����ͬϵ�� �������顢�����顢������ķе����
�ݱ�״���£�11.2 L�ļ��������ķ�����Ϊ0.5NA(NAΪ�����ӵ�����)
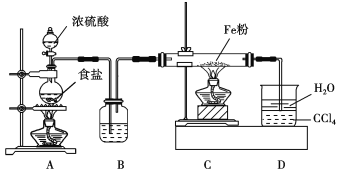
���ڶ��ױ���Һ���ڹ�����ȡ���������Ϊ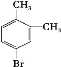
����ϩ�ͱ�����ϩ�ͱ��зֱ��������KMnO4��Һ�����۲��Ƿ���ɫ
A. �٢ڢۢܢ�B. �ڢۢܢ�C. �ڢۢ�D. �ۢݢ�
���𰸡�A
��������
��CH3-CH=CH2��CH2=CH2�����ʽ��ΪCH2����ͬ���ʢ���ȷ��
�����ʽ��ΪCH����C����ͬ����Ϊ92.3%���ʢ���ȷ��
��̼ԭ������ͬ��ֱ���������ṹ���ƣ�����������n��CH2����Ϊͬϵ��ʢ���ȷ��
����ͬCԭ�Ӹ�����������֧��Խ�࣬�е�Խ�ͣ��������顢�����顢������ķе���ͣ��ʢ���ȷ��
�ݱ�״���£�����ΪҺ�壬��������������������ʵ������ʢݴ���
���ڶ��ױ���Һ���ڹ����·���ȡ����Ӧ��ȡ�������Ǽ��ϵ��ⱻ��ԭ��ȡ���������DZ����ϵ���ԭ�ӱ���ԭ����ȡ�����ʢ���
��ϩ��ʹ����KMnO4��Һ��ɫ����������ʹ����KMnO4��Һ��ɫ������������KMnO4��Һ��ϩ�ͱ����ʢ���ȷ��
�٢ڢۢܢ���ȷ���ʴ�ΪA��

 �㽭�¿γ���άĿ�������ʱ��ѵϵ�д�
�㽭�¿γ���άĿ�������ʱ��ѵϵ�д� ��������ϵ�д�
��������ϵ�д� ���ɶ���ܲ��¿�ֱͨ�߿�ϵ�д�
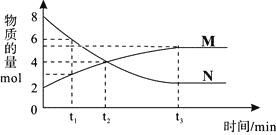
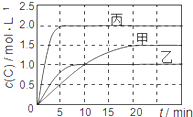
���ɶ���ܲ��¿�ֱͨ�߿�ϵ�д�����Ŀ����ס��ҡ��������ܱ������г���һ������A��B��������Ӧ��xA(g)��B(g)![]() 2C(g)���������ķ�Ӧ�¶ȡ���Ӧ����ʼ������Ӧ������C��Ũ����ʱ��仯��ϵ�ֱ����±�����ͼ��ʾ��
2C(g)���������ķ�Ӧ�¶ȡ���Ӧ����ʼ������Ӧ������C��Ũ����ʱ��仯��ϵ�ֱ����±�����ͼ��ʾ��
���� | �� | �� | �� |
�ݻ� | 1L | 1L | 2L |
�¶� | T1 | T2 | T2 |
��Ӧ�� ��ʼ�� | 1molA 2molB | 1molA 2molB | 4molA 8molB |
����˵����ȷ���� �� ��
A. ��ͼ��֪��T1��T2���Ҹ÷�ӦΪ���ȷ�Ӧ
B. T1ʱ�÷�Ӧ��ƽ�ⳣ��K=7.2
C. ǰ10min�ס��ҡ�������������A��ƽ����Ӧ���ʣ�v(A)����v(A)����v(A)��
D. ƽ��ʱA������m��m����m����m��
����Ŀ����֪��Ӧ��N2(g)��3H2(g)=2NH3(g)���й����ʵĻ�ѧ������(��1 mol��ѧ������Ҫ������)���£�
��ѧ�� | N��N | N��H | H��H |
����(kJ/mol) | 945.0 | 391.0 | 436.0 |
����÷�Ӧ����2 mol NH3ʱ���ջ�ų��������Ƕ��٣�