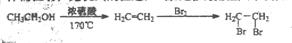��Ŀ����
��ѧ�Ҽ�һЩʵҵ�ҵĹ��Կ�ѧ�ķ�չ���Ż������ƶ����á����ж��йؿ�ѧ�Ҽ��䷢��������������������
| A��������֤���˻�ѧ��Ӧ�е������غ㣬������ȼ�յ������� |
| B�����շ���������������������˱��Ľṹʽ |
| C����°����ˡ������Ƽ�������̳��������й����ȼҵ |
| D�������١���ķ�¬ɪ�����Ž��з�ȿ�ѧ�Ҷ�ԭ�ӽṹģ�͵Ľ�����������Խ�Ĺ��� |
D
����������Ž��з�ΪԪ�����ڱ������ߡ���Dѡ�����

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
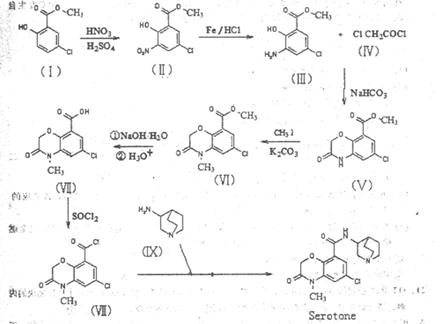
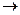 ����NaHCO3gm V
����NaHCO3gm V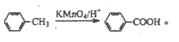
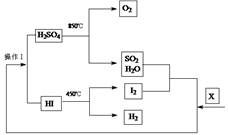
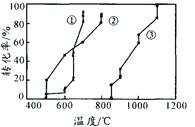
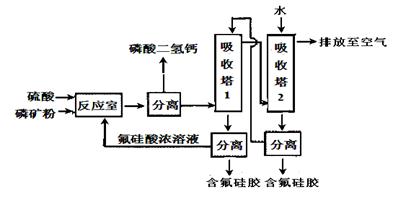
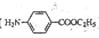 ���ĺϳ�·������ͼ����Ҫ������ע�������Լ���ѡ�����ϳ�·������ͼʾ�����£�
���ĺϳ�·������ͼ����Ҫ������ע�������Լ���ѡ�����ϳ�·������ͼʾ�����£�