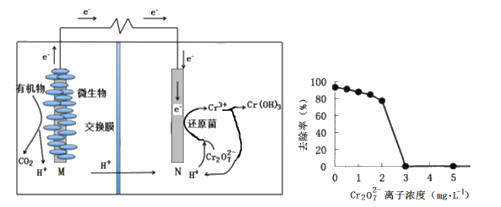
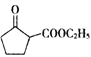
题目内容
【题目】(1)如图所示,将氯气依次通过盛有干燥有色布条的广口瓶和盛有潮湿有色布条的广口瓶,可观察到的现象是 ______________。

(2)为防止氯气尾气污染空气,可用________溶液吸收多余的氯气,原理是(用化学方程式表示)____________________。
(3)工业上常用廉价的石灰乳吸收工业氯气尾气制得漂白粉,漂白粉的有效成分是_______(填化学式),长期露置于空气中的漂白粉,加稀盐酸后产生的气体是_______(用字母代号填)。
A.O2 B.Cl2 C.CO2 D.HClO
【答案】干燥的有色布条无明显现象,潮湿的有色布条褪色 NaOH Cl2+2NaOH=NaCl+NaClO+H2O Ca(ClO)2 C
【解析】
本题考查次氯酸的漂白性,氯气与碱的反应以及漂白粉失效的原理。回答时要注意语言的规范和严谨。
(1)干燥的氯气没有漂白作用,潮湿的氯气,因为氯气与水反应生成了次氯酸而具有漂白作用。所以,将氯气依次通过盛有干燥有色布条的广口瓶和盛有潮湿有色布条的广口瓶,可观察到的现象是:干燥的有色布条无明显现象,潮湿的有色布条褪色;
(2)为防止尾气中的氯气污染空气,通常用浓NaOH溶液进行吸收,反应的化学方程式为:Cl2+2NaOH=NaCl+NaClO+H2O;
(3)工业上常用廉价的石灰乳吸收工业氯气尾气制得漂白粉:2Cl2+2Ca(OH)2=Ca(ClO)2+CaCl2+2H2O,漂白粉的主要成分是Ca(ClO)2和CaCl2,有效成分是Ca(ClO)2。漂白粉长期露置于空气中会与CO2和水反应:Ca(ClO)2+CO2+H2O=CaCO3+2HClO,所得固体中加稀盐酸后发生反应:CaCO3+2HCl=CaCl2+H2O+CO2↑,产生的气体主要是CO2气体。

【题目】温度为T时,向2.0 L恒容密闭容器中充入1.0 mol PCl5,发生反应PCl5(g)![]() PCl3(g)+Cl2(g),经过一段时间后达到平衡。反应过程中测定的部分数据见下表,下列说法正确的是
PCl3(g)+Cl2(g),经过一段时间后达到平衡。反应过程中测定的部分数据见下表,下列说法正确的是
t / s | 0 | 50 | 150 | 250 | 350 |
n(PCl3) / mol | 0 | 0. 16 | 0. 19 | 0. 20 | 0. 20 |
A. 反应在前50 s 的平均速率ν(Cl2)= 0. 0032 mol·L-1·s-1
B. 保持其他条件不变,升高温度,平衡时c(PCl3)= 0. 13 mol·L-1,则反应的ΔH<0
C. 相同温度下,起始时向容器中充入1. 0 mol PCl5、0. 20 mol PCl3 和0. 20 mol Cl2 ,反应达到平衡前ν(正)<ν(逆)
D. 相同温度下,起始时向容器中充入2. 0 mol PCl3 和2. 0 mol Cl2 ,达到平衡时,PCl3 的转化率大于80%